1, 2 - Radiology department, Kasr Alainy hospitals, Cairo University, Egypt;
3 - Fetal medicine unit, Cairo University, Egypt.
Case report
29-year-old woman (G1P0) was referred to our office at 27 weeks gestation due to abnormal cardiac anatomy.
Our ultrasound examination revealed:
-
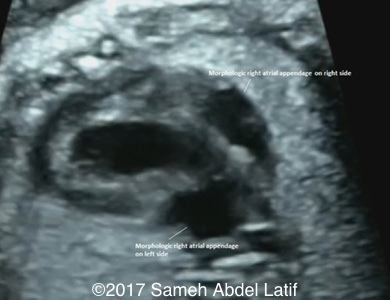
Situs ambiguous (Central liver, left sided stomach, T-shaped umbilico-portal connection and juxta posed descending aorta and IVC). Both atrial appendages were of right sided morphology.
-
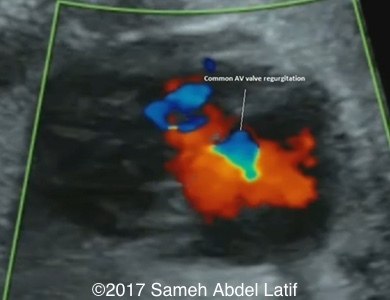
Single ventricle with common AV canal showing mild regurgitation across.
-
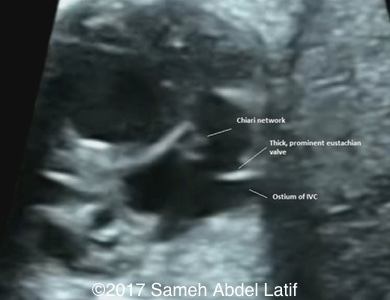
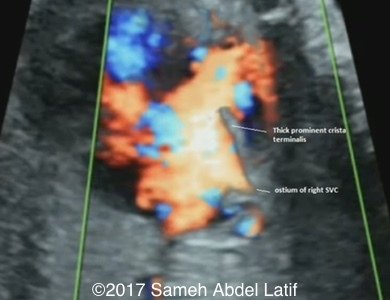
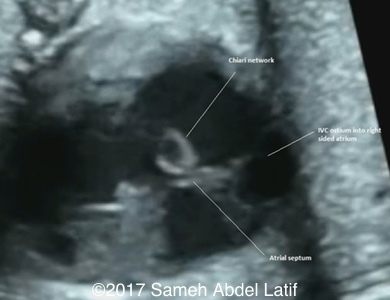
Drainage of the pulmonary veins and the both SVCs into left sided atrium while the IVC drains into the right sided atrium. No bridging vein was noted between both SVCs. Chiari network with prominent Crista terminalis and Eustachian valve were also noted in the right sided atrium.
-
Single outlet morphology. The single ventricle gives one artery that forms the aortic arch.
-
Hypoplastic, confluent both central pulmonary arteries. Each measures 4 mm in maximum systolic dimension. Retrograde filling was seen through rather tortuous ductus arteriosus.
Images 1-6 and videos 1-6: show single ventricle, common AV valve with mild regurgitation across, both atrial appendages of right morphology, prominent crista terminalis, Eustachian valve and Chiari network in right sided atrium.
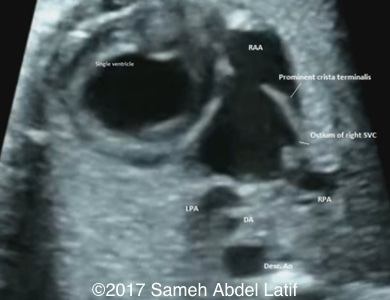
Images 7-14 and videos 7-12: show single arterial outlet (ascending aorta), pulmonary atresia with central confluent branch pulmonary arteries and retrograde filling through ductus arteriosus and bilateral SVC draining into left sided atrium that also receives pulmonary veins.
Images 15-18 and videos 13-15: show central liver, juxta posed descending aorta and IVC, T-shaped appearance of umbilico-portal connection (dilated esophagus is noted but not persistent during whole time of scan).
Images 19, 20 and videos 16, 17: Volume rendered images and videos showing ductus arteriosus arising from isthmic portion of aortic arch to end at the base of LPA, both central, confluent pulmonary arteries appear Hypoplastic and completely dependent upon the aorta.
Images 21-26, videos 18 and 19: Pulmonary arterial flow mapping by STIC technique during late diastolic and whole systolic phases showing relatively delayed filling of right pulmonary artery than the left one confirming their central confluence and excluding origin of RPA from the single trunk (aorta).
Our final diagnosis was pulmonary atresia with complete duct dependent pulmonary circulation and associated single ventricle, common AV canal and bilateral SVCs with right atrial isomerism.We recommended immediate postnatal initiation of prostaglandin therapy as a lifesaving maneuver (as tortuous ductus arteriosus pathway makes it difficult to be stented) and then urgent MBT shunt to enhance growth of pulmonary arteries prior to single ventricle repair procedures however, 4 days postnatal, the baby suffered from central cyanosis and died despite intensive resuscitation measures.
Comment
In our case, some imaging findings deserve comment. It is of concern that this anomaly may represent type III common arterial trunk (CAT) according to Van Praagh classification in which the left pulmonary artery arises from the ductus arteriosus while the right sided one arises by a separate ostium from the posterior wall of the trunk.
However, we found the confluence of the hypoplastic central pulmonary arteries just caudal to the ascending aorta with normal looking of aortic valve leaflets. Using STIC technique, it was much more helpful in confirming our diagnosis as pulmonary atresia with duct dependent pulmonary circulation than type III CAT. There was time delay between early systolic retrograde filling of the left pulmonary artery and mid and late systolic filling of right pulmonary artery. On the other hand, there will be no time delay between both pulmonary arteries in case of type III CAT. This finding is extremely important to differentiate between critical condition (pulmonary atresia) and a condition that could be managed conservatively till proper timing of corrective surgery.
Color STIC assisted flow mapping of pulmonary arteries and aorta was very helpful in confirming diagnosis and thus could be an additional tool to evaluate confluence of central pulmonary arteries in case of pulmonary atresia and to differentiate it from the non-critical anomaly (type III common arterial trunk).
References
-
Crupi, G., Macartney, F. J., & Anderson, R. H. (1977). Persistent truncus arteriosus: a study of 66 autopsy cases with special reference to definition and morphogenesis. The American journal of cardiology, 40(4), 569-578.
-
Słodki, M., Respondek-Liberska, M., Pruetz, J. D., & Donofrio, M. T. (2016). Fetal cardiology: changing the definition of critical heart disease in the newborn. Journal of Perinatology, 36(8), 575-580.