Case of the Week #648
(1) Femicare, Center of prenatal ultrasonographic diagnostics, Martin, Slovakia; (2) Centro Médico Recoletas, Valladolid, Spain
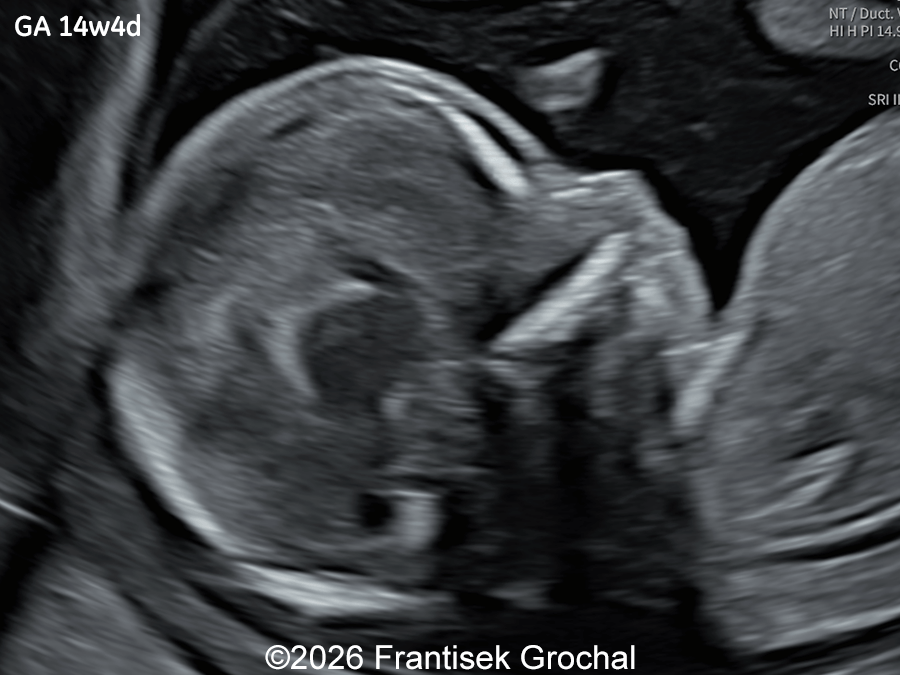
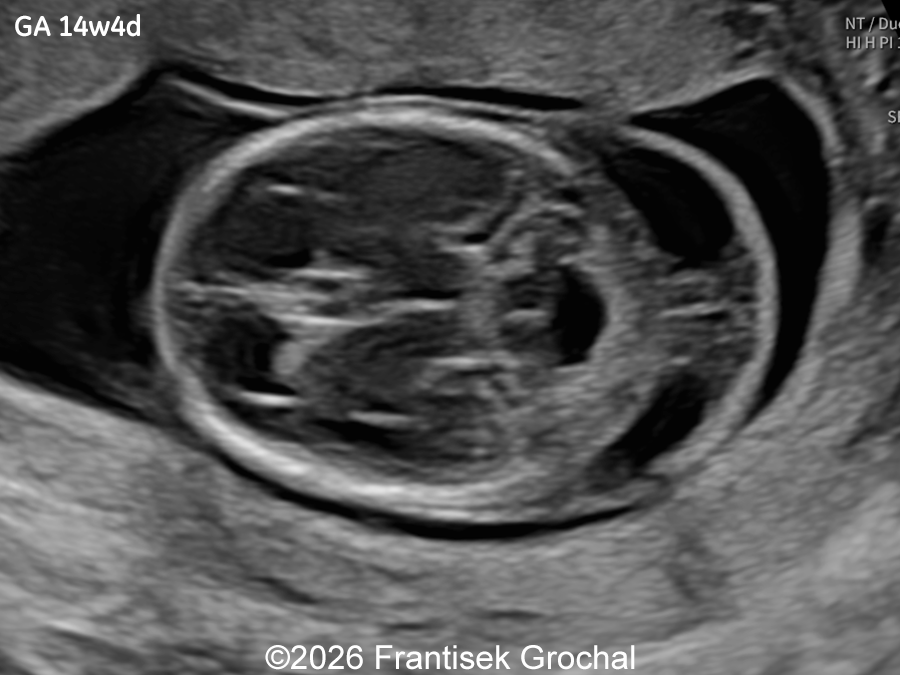
A 30-year-old primigravida presented to our outpatient clinic for ultrasound examinations at 14 and subsequently at 19 weeks of gestation. In a male fetus, the following findings were identified:
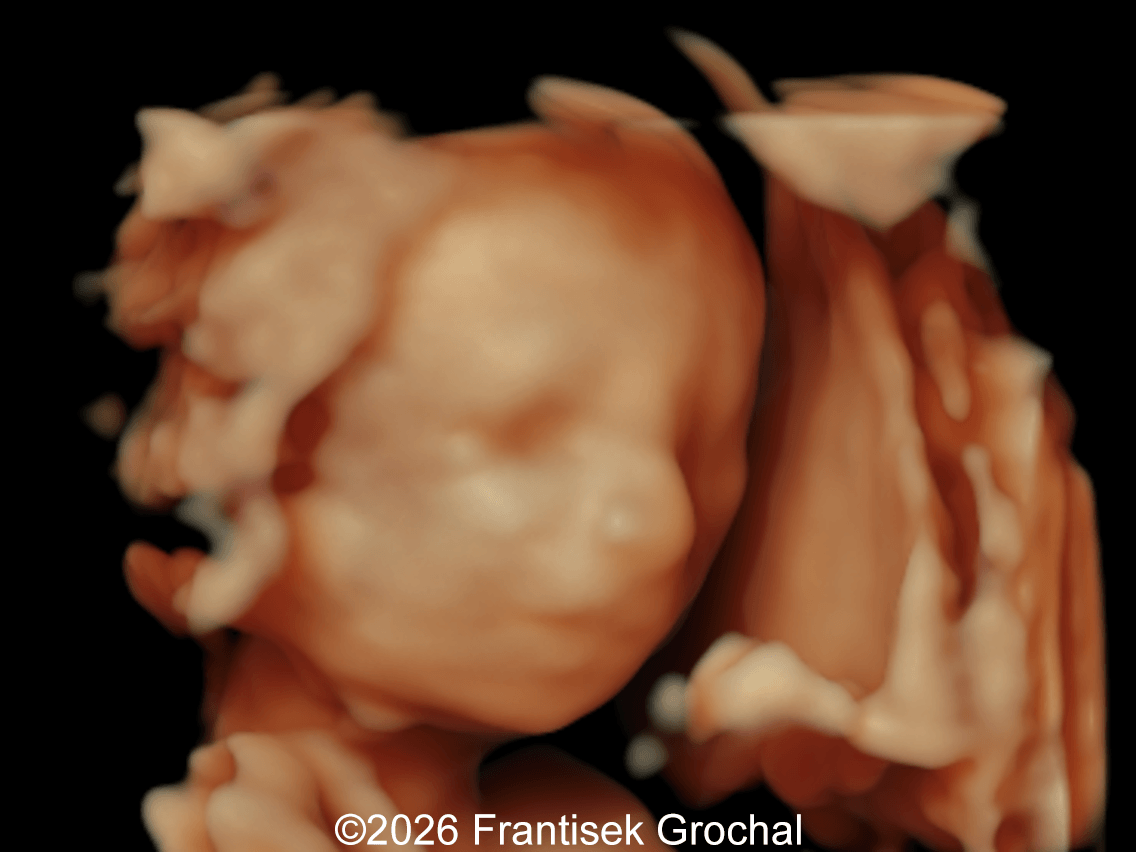
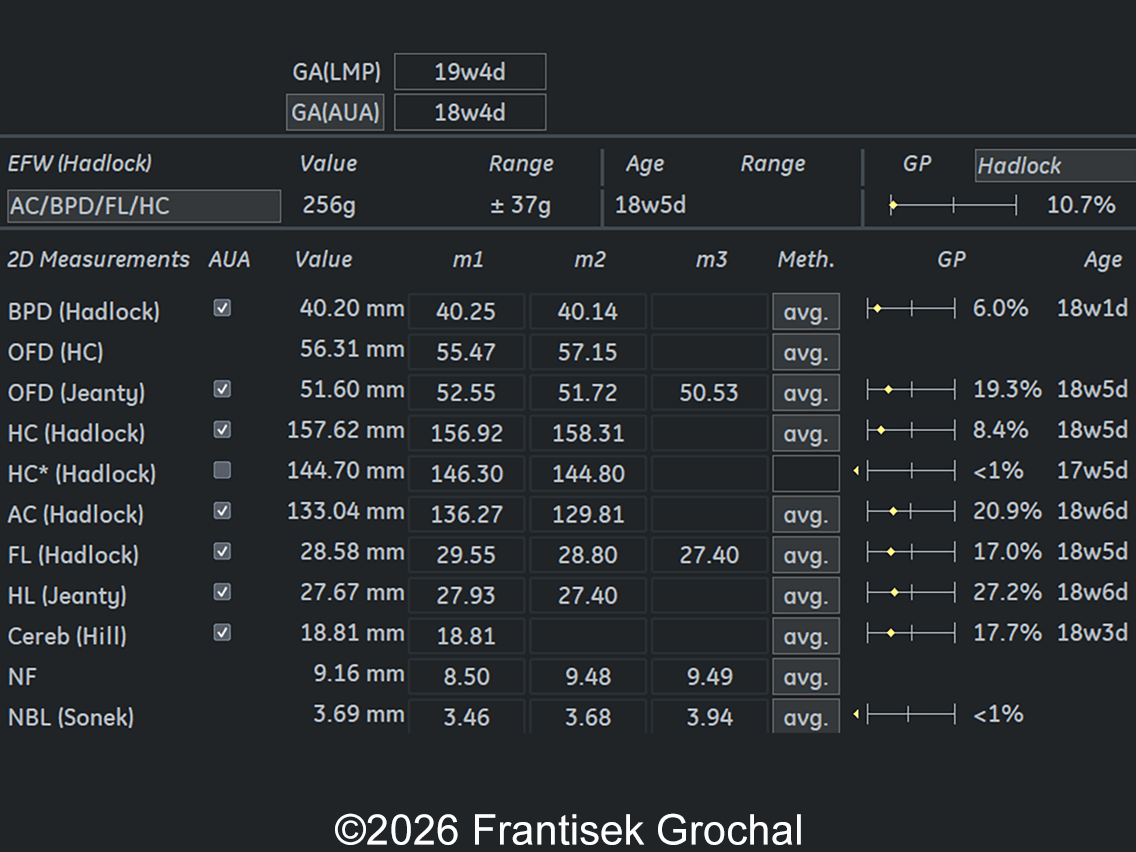
View the Answer Hide the Answer
Answer
We present a case of Wolf–Hirschhorn syndrome.
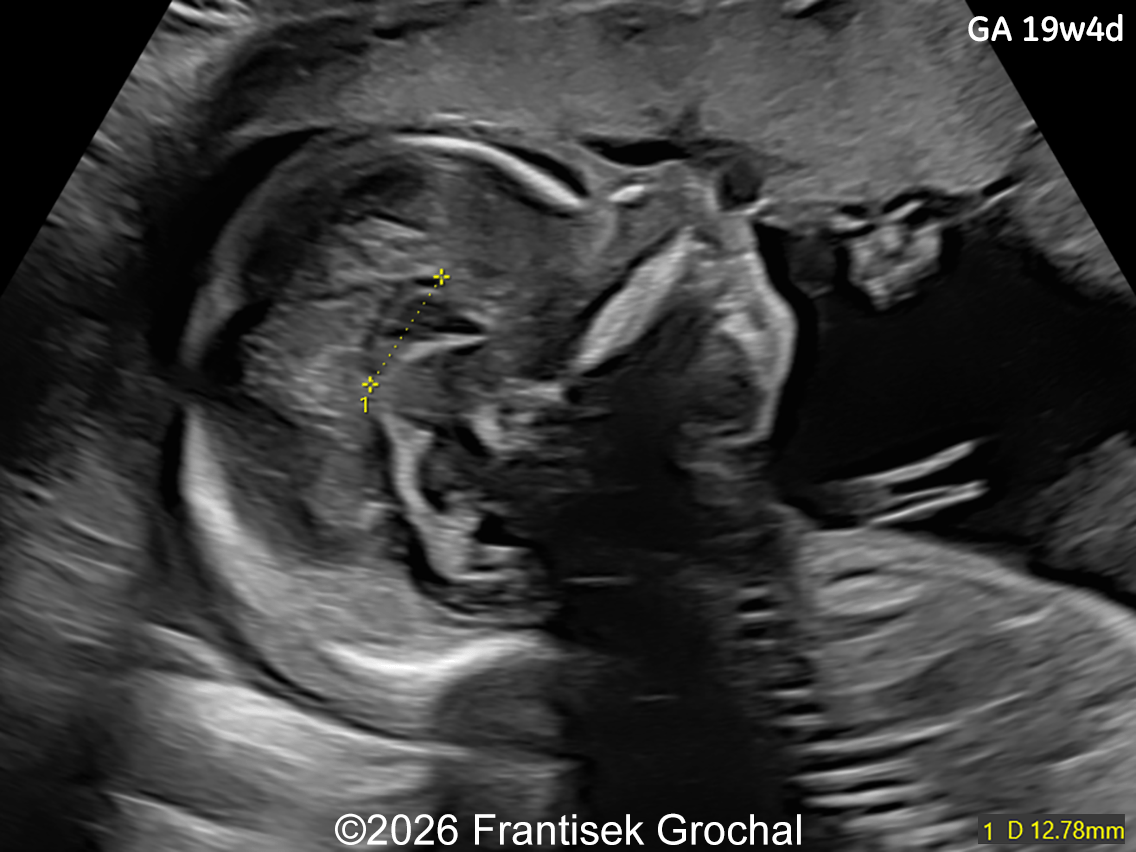
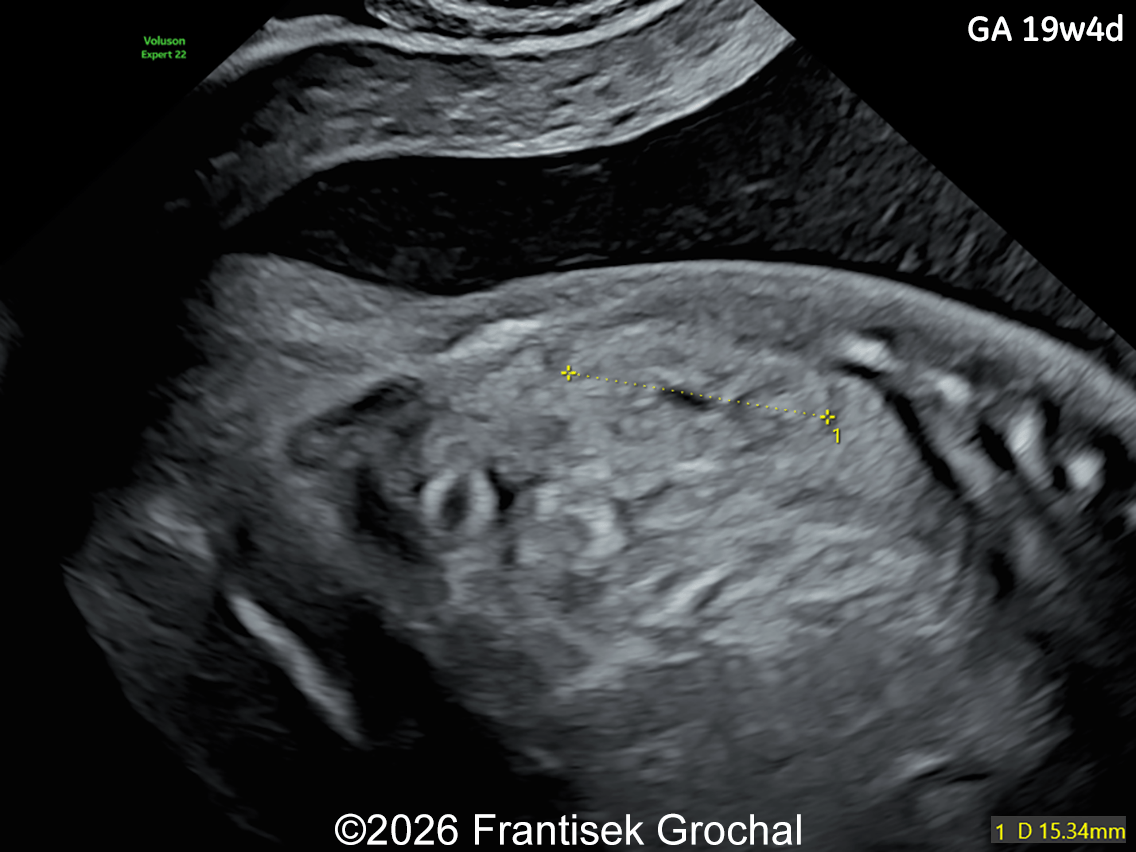
Ultrasound examination at 14 weeks demonstrated marked nuchal edema, nasal bone hypoplasia, and a suspected ventricular septal defect.
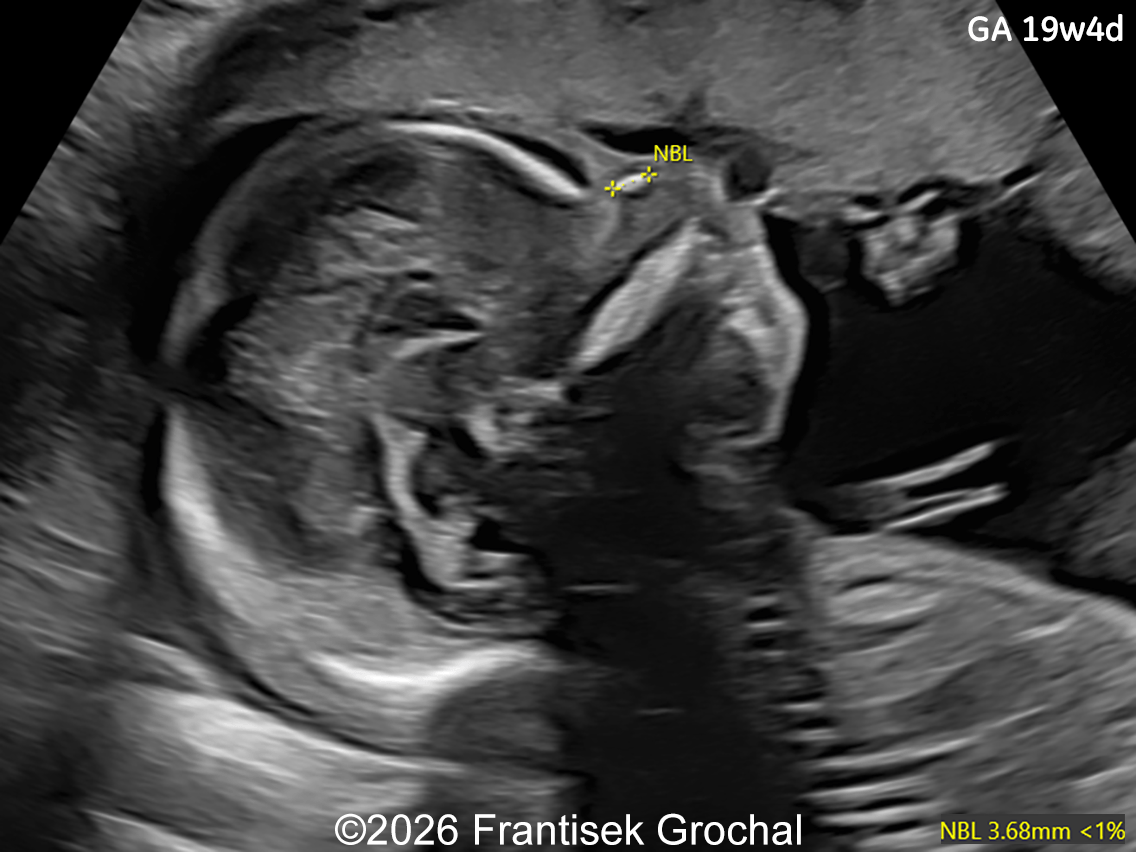
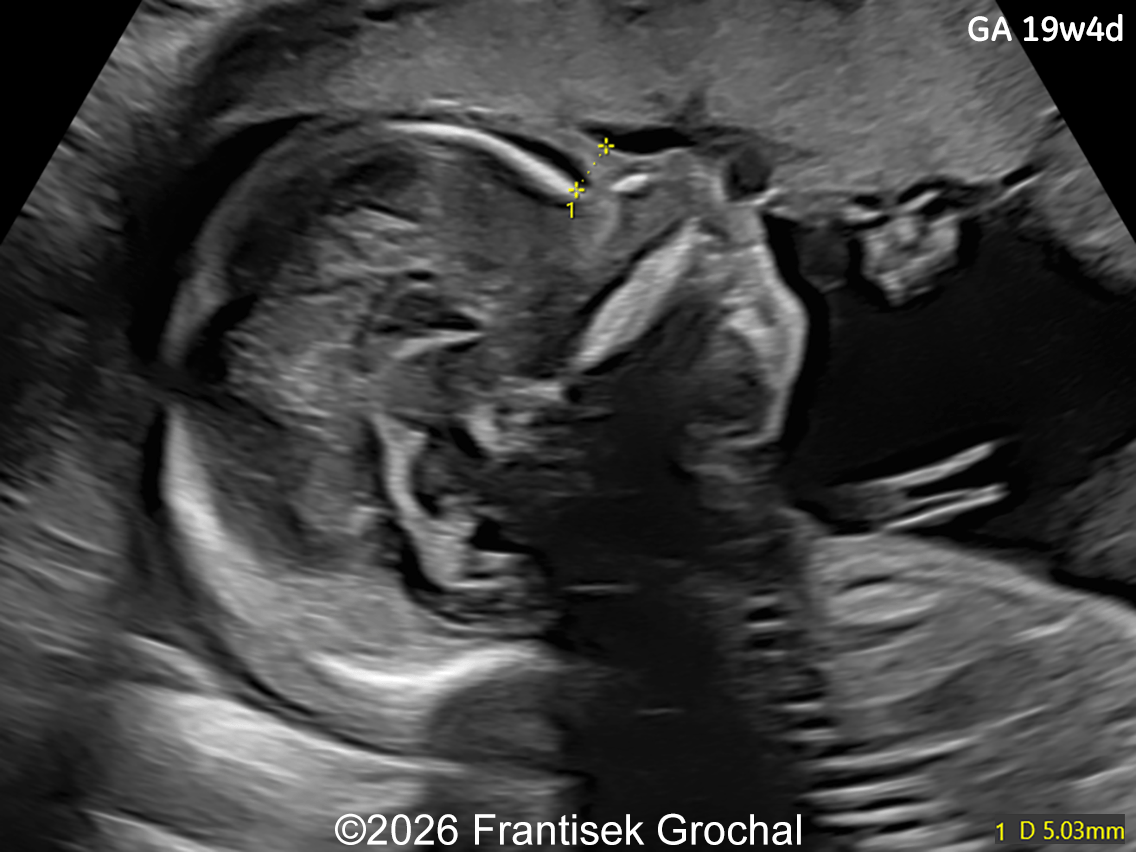
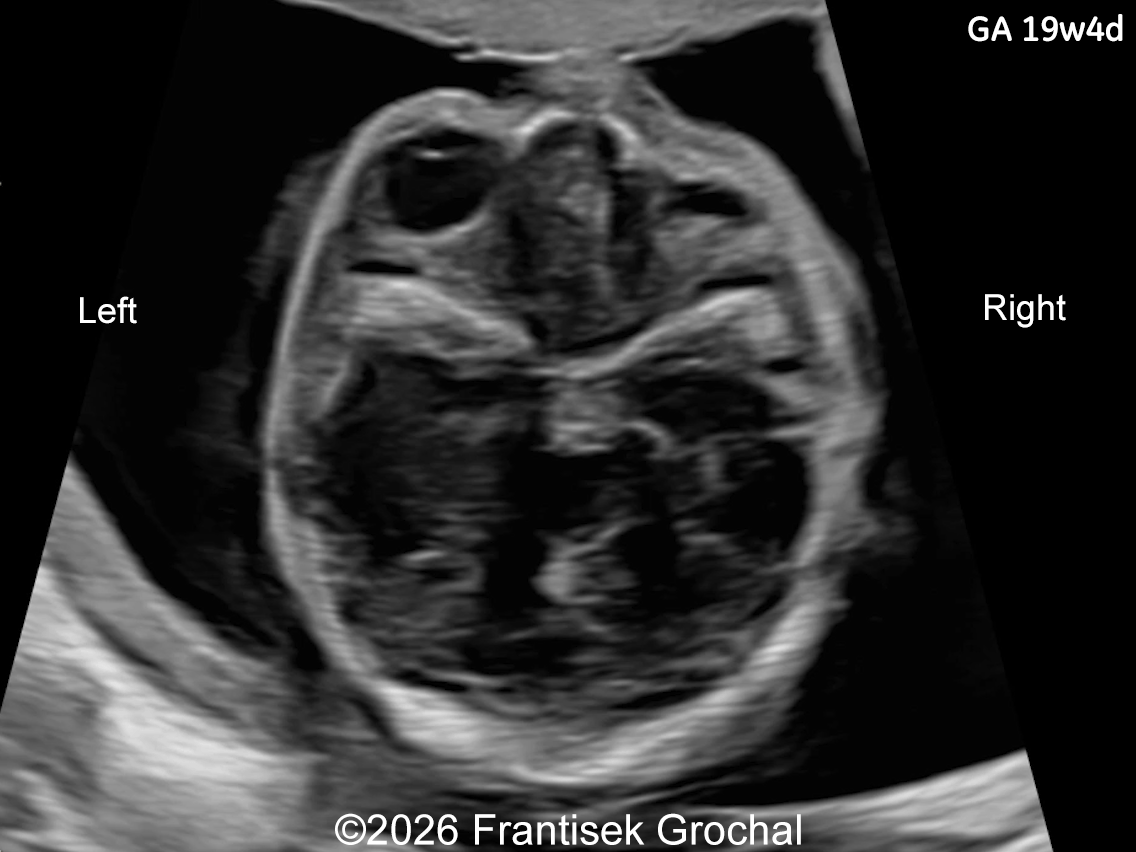
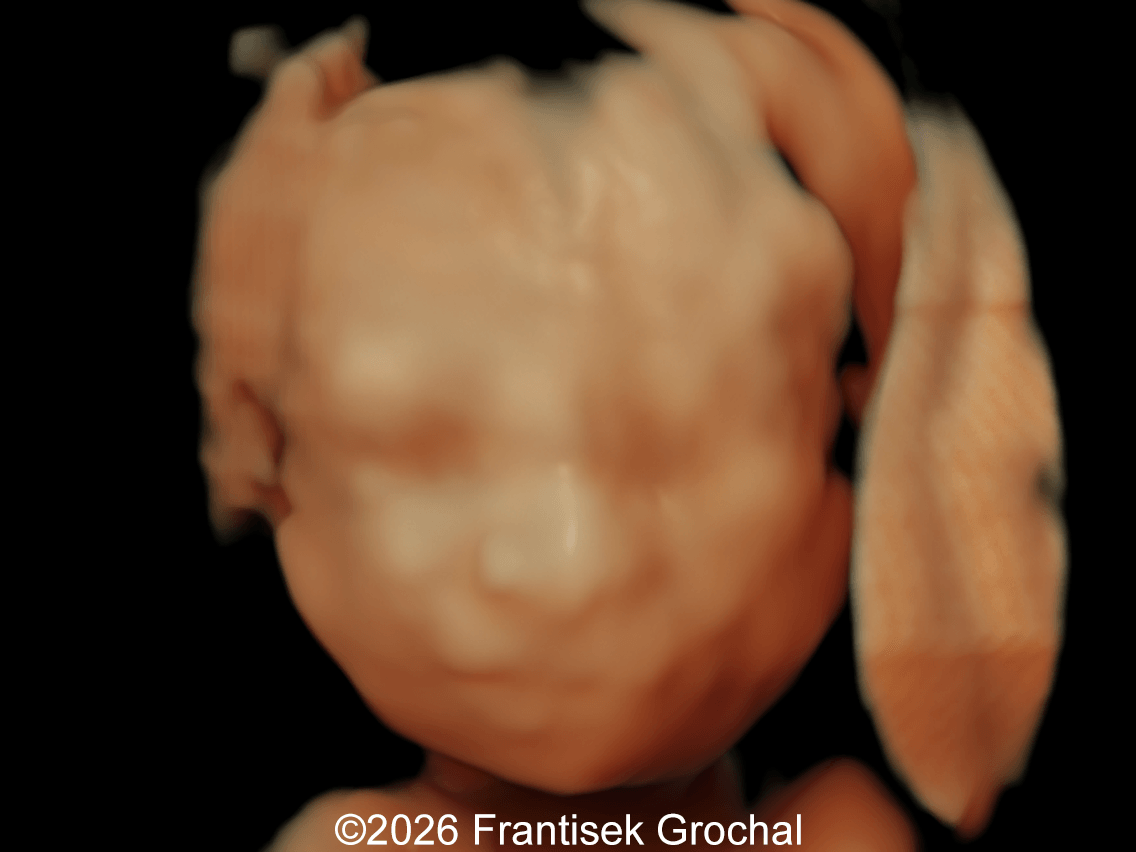
Follow-up examination at 19 weeks revealed multiple structural abnormalities:
- Marked nuchal edema
- Prefrontal edema
- Nasal bone hypoplasia
- Micrognathia
- Right-sided microphthalmia
- Short corpus callosum
- Ventricular septal defect (VSD)
- Persistent right subclavian artery (ARSA)
- Renal hypoplasia
- Fetal growth restriction, with biometric parameters lagging 7 days behind the gestational age calculated according to the last menstrual period
Given the constellation of anomalies, a chromosomal abnormality was suspected. Amniocentesis was performed, and genetic testing confirmed Wolf–Hirschhorn syndrome. After multidisciplinary counseling regarding the prognosis, the parents opted for termination of pregnancy.

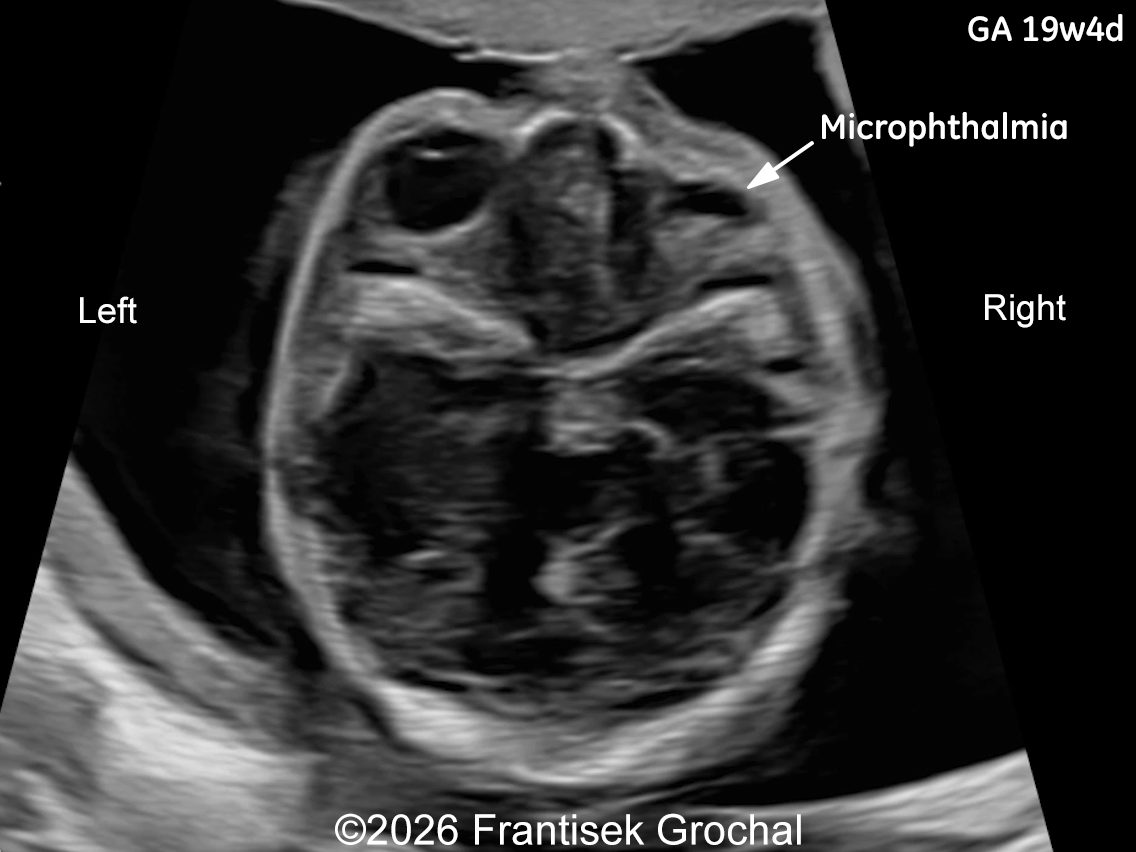
Discussion
Wolf–Hirschhorn syndrome (OMIM # 194190), also known as Pitt-Rogers-Danks syndrome, is a chromosomal disorder caused by a distal deletion of the short arm of chromosome 4, with loss of the terminal band 4p16.3 which is essential for the full expression of the phenotype [1]. The clinical picture was first described in 1961 by Cooper and Hirschhorn in a child with midline fusion defects, low birth weight, developmental delay, and neonatal-onset seizures [2]. Four years later, Wolf et al. reported a second patient with similar features [3]. These two reports established the existence of human syndromes caused by chromosomal deletions, of which Wolf–Hirschhorn syndrome represents the first recognized example. Its estimated incidence of 1 in 50,000 live births is derived from standard karyotyping, which detects only large 4p16.3 deletions. The introduction of FISH, and later genomic microarrays, improved the detection of small deletions and milder phenotypes, suggesting that historical numbers are likely underestimated and incidence of the syndrome may be 1 in 20,000 [4,5].
Two Wolf–Hirschhorn syndrome critical regions (WHSCR and WHSCR-2) within 4p16.3 contain multiple genes, including WHSC1 (NSD2), LETM1, CPLX1, FGFRL1, and PIGG, which have been investigated for their contribution to different clinical features of the syndrome [4,6-9]. Haploinsufficiency of WHSC1 has been proposed to contribute to the characteristic craniofacial features of the syndrome, whereas hemizygosity of LETM1 has been associated with the occurrence of seizures. However, current evidence indicates that neither gene alone is sufficient to account for these phenotypes, and the contribution of these loci to growth retardation remains inconsistent and somewhat conflicting [8]. As the size of the detectable deletion decreases, the number of associated traits correspondingly declines, supporting the notion that Wolf-Hirschhorn syndrome is a true contiguous gene syndrome rather than the result of a single pleiotropic gene deletion [7]. Genomic rearrangements are mostly de novo (including isolated deletions, unbalanced translocations, inverted duplications, and unbalanced pericentric inversions) but can originate from a balanced parental translocation in 10% to 15% of cases [9]. De novo arrangement tends to be paternal in origin [10]. Due to this genetic heterogeneity, there is significant clinical variability as well. Parents who carry balanced translocations involving 4p16.3 have an increased risk of having offspring with an inherited unbalanced translocation.
There is broad consensus that the postnatal diagnosis of Wolf-Hirschhorn syndrome relies on the association of characteristic facial dysmorphism (prominent forehead, hypertelorism, and wide nasal bridge continuing to the forehead, known as “Greek warrior helmet appearance”), growth retardation, intellectual disability, and seizures or electroencephalographic abnormalities [8]. Zollino et al [8] suggested classification of the syndrome based on the size of the deletion: “mild” (deletions < 3.5 Mb), “classical” (deletions 5–18 Mb), and “severe” forms (deletions > 22–25 Mb).
Most prenatally diagnosed cases show growth restriction, microcephaly, and characteristic craniofacial appearance. The severe early‑onset growth restriction is present in nearly all affected fetuses [11,12] and marked microcephaly persists after birth [13]. The characteristic craniofacial appearance is defined by a prominent glabella, wide nasal bridge with a beaked nose, high forehead, hypertelorism, downturned corners of the mouth, and a short philtrum [14-16]. Based on combined pre‑ and postnatal data, the ultrasound phenotype can be grouped according to its frequency [Table 1] [1]:
| Frequency of findings | Ultrasound phenotype |
|---|---|
| Findings present >75% |
|
Findings present in 50–75% |
|
Findings present in 25–50% |
|
| Findings present in <25% |
|
Occasionally, Wolf–Hirschhorn syndrome has been diagnosed in the first trimester following the finding of increased nuchal translucency or a cystic hygroma [27,31-33]. Although these early sonographic features are non‑specific and have a wide differential diagnosis, they warrant invasive testing which leads to the diagnosis.
In our case of prenatally suspected Wolf–Hirschhorn syndrome, diagnosis was made based on characteristic sonographic findings and confirmed by invasive genetic testing. Increased nuchal edema, early fetal growth restriction, and the coexistence of multiple structural abnormalities should raise suspicion for an underlying genetic disorder. Our observations are consistent with previously published prenatal phenotypes of Wolf–Hirschhorn syndrome and support the role of systematic neurosonographic and cardiac evaluation when multiple minor abnormalities are detected. This case highlights the importance of detailed first- and second-trimester ultrasound assessment in detecting complex chromosomal syndromes.
The prenatal differential diagnosis of Wolf–Hirschhorn syndrome is broad, reflecting the nonspecific nature of its early sonographic findings, most commonly increased nuchal translucency, cystic hygroma, early‑onset fetal growth restriction, and multiple congenital anomalies. These features overlap with several chromosomal and genetic conditions, including trisomy 18 (Edwards syndrome), 11q deletion syndrome (Jacobsen syndrome), Fryns syndrome (particularly when a diaphragmatic hernia is present), Seckel syndrome, VACTERL association, CHARGE syndrome, and Smith–Lemli–Opitz syndrome [1]. This overlap underscores the need for comprehensive evaluation through invasive testing and chromosomal microarray analysis.
References
- Adams A, Lee W, and Van den Veyver IB. Chromosome 4p Deletion Syndrome (Wolf-Hirschhorn Syndrome). In: Copel JA, ed. Obstetric Imaging. Fetal diagnosis and care, 3rd ed. Elsevier, Philadelphia, PA, 2026; pag 688-692.e1.
- Hirschhorn K, Cooper HL. 1961. Apparent deletion of short arms of one chromosome (4 or 5) in a child with defects of midline fusion. Human Chromosome Newslett IV:14.
- Wolf U, Reinwein H, Porsch R, Schröter R. Baitsch H. 1965. Defizienz an den kurzen Armen eines Chromosoms Nr. 4. Humangenetik 1:397.
- Maas NM, Van Buggenhout G, Hannes F, et al. Genotype-phenotype correlation in 21 patients with Wolf-Hirschhorn syndrome using high resolution array comparative genome hybridisation (CGH). J Med Genet. 2008 Feb;45(2):71-80.
- Battaglia A, Carey JC, South ST. Wolf-Hirschhorn syndrome: A review and update. Am J Med Genet C Semin Med Genet. 2015 Sep;169(3):216-223.
- Gandelman KY, Gibson L, Meyn MS, Yang-Feng TL. Molecular definition of the smallest region of deletion overlap in the Wolf-Hirschhorn syndrome. Am J Hum Genet. 1992 Sep;51(3):571-578.
- Estabrooks LL, Rao KW, Driscoll DA, et al. Preliminary phenotypic map of chromosome 4p16 based on 4p deletions. Am J Med Genet. 1995 Jul 17;57(4):581-586.
- Zollino M, Murdolo M, Marangi G, et al. On the nosology and pathogenesis of Wolf-Hirschhorn syndrome: genotype-phenotype correlation analysis of 80 patients and literature review. Am J Med Genet C Semin Med Genet. 2008 Nov 15;148C(4):257-269.
- Nevado J, Ho KS, Zollino M, et al. International meeting on Wolf-Hirschhorn syndrome: Update on the nosology and new insights on the pathogenic mechanisms for seizures and growth delay. Am J Med Genet A. 2020 Jan;182(1):257-267.
- Quarrell OW, Snell RG, Curtis MA, et al. Paternal origin of the chromosomal deletion resulting in Wolf-Hirschhorn syndrome. J Med Genet. 1991 Apr;28(4):256-259.
- Tachdjian G, Fondacci C, Tapia S, et al. The Wolf-Hirschhorn syndrome in fetuses. Clin Genet. 1992 Dec;42(6):281-287.
- Xing Y, Holder JL Jr, Liu Y, et al. Prenatal diagnosis of Wolf-Hirschhorn syndrome: from ultrasound findings, diagnostic technology to genetic counseling. Arch Gynecol Obstet. 2018 Aug;298(2):289-295.
- Simonini C, Hoopmann M, Kagan KO, et al. Prenatal sonographic findings in confirmed cases of Wolf-Hirschhorn syndrome. BMC Pregnancy Childbirth. 2022 Apr 15;22(1):327.
- Levaillant JM, Touboul C, Sinico M, et al. Prenatal forehead edema in 4p- deletion: the 'Greek warrior helmet' profile revisited. Prenat Diagn. 2005 Dec;25(12):1150-1155.
- Sepulveda W. Prenatal 3-dimensional sonographic depiction of the Wolf-Hirschhorn phenotype: the "Greek warrior helmet" and "tulip" signs. J Ultrasound Med. 2007 Mar;26(3):407-410.
- Hanprasertpong T, Hanaoka U, Zhang X, et al. Three-dimensional sonographic features of a fetus with Wolf-Hirschhorn syndrome. J Med Ultrason (2001). 2008 Dec;35(4):197-199.
- Dietze I, Fritz B, Huhle D, et al. Clinical, cytogenetic and molecular investigation in a fetus with Wolf-Hirschhorn syndrome with paternally derived 4p deletion. Case report and review of the literature. Fetal Diagn Ther. 2004 May-Jun;19(3):251-260.
- Boog G, Le Vaillant C, Collet M, et al. Prenatal sonographic patterns in six cases of Wolf-Hirschhorn (4p-) syndrome. Fetal Diagn Ther. 2004 Sep-Oct;19(5):421-430.
- von Elten K, Sawyer T, Lentz-Kapua S, et al. A case of Wolf-Hirschhorn syndrome and hypoplastic left heart syndrome. Pediatr Cardiol. 2013 Jun;34(5):1244-1246.
- Beaujard MP, Jouannic JM, Bessières B, et al. Prenatal detection of a de novo terminal inverted duplication 4p in a fetus with the Wolf-Hirschhorn syndrome phenotype. Prenat Diagn. 2005 Jun;25(6):451-455.
- Sifakis S, Manolakos E, Vetro A, et al. Prenatal diagnosis of Wolf-Hirschhorn syndrome confirmed by comparative genomic hybridization array: report of two cases and review of the literature. Mol Cytogenet. 2012 Feb 28;5:12.
- Bonasoni MP, Pati M, Shah K, et al. Renal Hypoplasia and Oligomeganephronia in a Fetus with Wolf-Hirschhorn Syndrome. Diagnostics (Basel). 2025 Oct 24;15(21):2687.
- Viñals F, Sepulveda W, Selman E. Prenatal detection of congenital hypospadias in the Wolf-Hirschhorn (4p-) syndrome. Prenat Diagn. 1994 Dec;14(12):1166-1169.
- Aslan H, Karaca N, Basaran S, et al. Prenatal diagnosis of Wolf-Hirschhorn syndrome (4p-) in association with congenital hypospadias and foot deformity. BMC Pregnancy Childbirth. 2003 Jan 24;3(1):1.
- Ikonomou T, Antsaklis P, Daskalakis G, et al. Prenatal diagnosis of Wolf-Hirschhorn syndrome: ultrasonography and genetics. J Matern Fetal Neonatal Med. 2013 Jun;26(9):941-942.
- Verbrugge J, Choudhary AK, Ladda R. Tethered cord, corpus callosum abnormalities, and periventricular cysts in Wolf-Hirschhorn syndrome. Report of two cases and review of the literature. Am J Med Genet A. 2009 Oct;149A(10):2280-2284.
- Basgul A, Kavak ZN, Akman I, et al. Prenatal diagnosis of Wolf-Hirschhorn syndrome (4p-) in association with congenital diaphragmatic hernia, cystic hygroma and IUGR. Clin Exp Obstet Gynecol. 2006;33(2):105-106.
- Casaccia G, Mobili L, Braguglia A, et al. Distal 4p microdeletion in a case of Wolf-Hirschhorn syndrome with congenital diaphragmatic hernia. Birth Defects Res A Clin Mol Teratol. 2006 Mar;76(3):210-213.
- Tautz J, Veenma D, Eussen B, et al. Congenital diaphragmatic hernia and a complex heart defect in association with Wolf-Hirschhorn syndrome. Am J Med Genet A. 2010 Nov;152A(11):2891-2894.
- Zhuang J, Huang N, Chen Y, et al. Prenatal diagnosis and genetic assessment of fetuses with single umbilical artery using chromosomal microarray analysis: a seven-year single-center retrospective study. BMC Pregnancy Childbirth. 2025 Jul 19;25(1):776.
- Verloes A, Schaaps JP, Herens C, et al. Prenatal diagnosis of cystic hygroma and chorioangioma in the Wolf-Hirschhorn syndrome. Prenat Diagn. 1991 Feb;11(2):129-132.
- Zhen L, Fan SS, Huang LY, et al. Prenatal diagnosis of Wolf-Hirschhorn syndrome: Ultrasonography and molecular karyotyping results. Eur J Obstet Gynecol Reprod Biol. 2018 Jun;225:19-21.
- Lin MF, Huang LY, Yang YD, Li DZ. Prenatal diagnosis of Wolf-Hirschhorn syndrome at the first trimester using chromosomal microarray analysis. J Obstet Gynaecol. 2019 Feb;39(2):268-270.
Discussion Board
Winners

Elena Andreeva Russian Federation Physician

Gulten Rafibeyli Azerbaijan Physician