Case of the Week #645
(1) Department of Radiology, Women' Yas Hospital, Tehran University of Medical Sciences, Tehran, Iran; (2) Centro Médico Recoletas, Valladolid, Spain
A 36-year-old pregnant woman (G2P1) was referred at 18 weeks and 4 days gestation to our tertiary center for sonographic evaluation due to the suspicion of various fetal anomalies.
View the Answer Hide the Answer
Answer
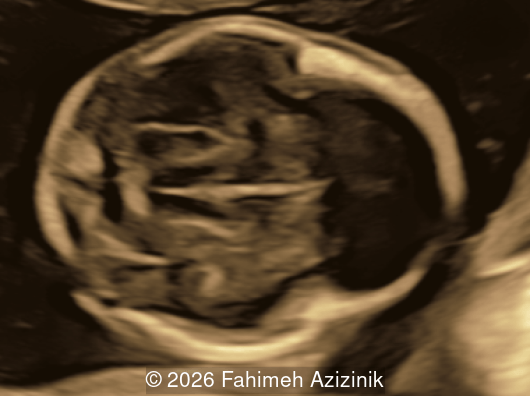
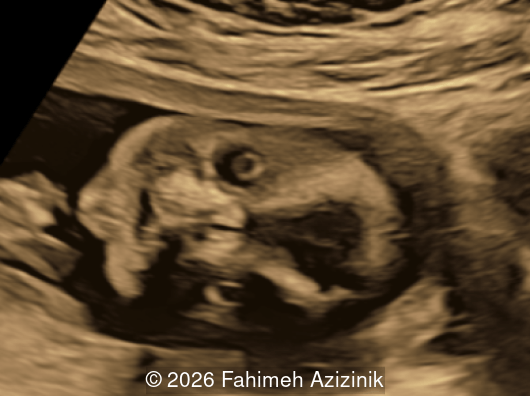
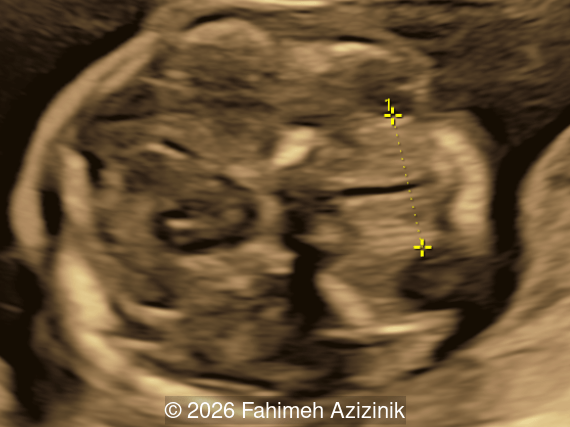
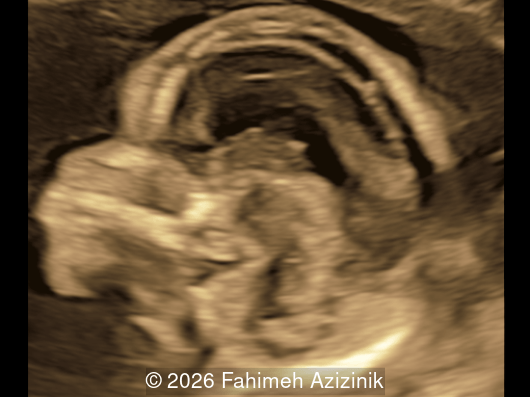
We present a case of Apert Syndrome.
Our ultrasound examination revealed the following findings:
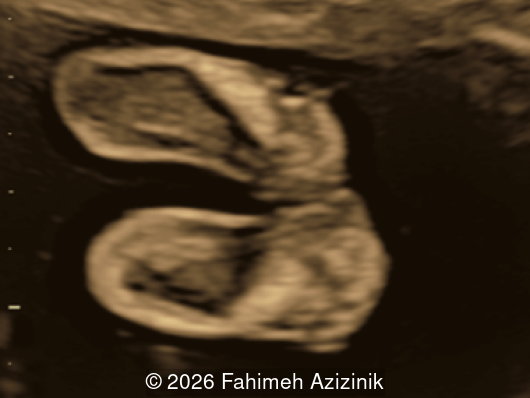
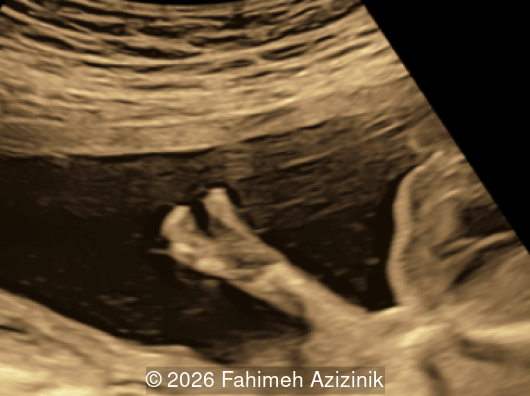
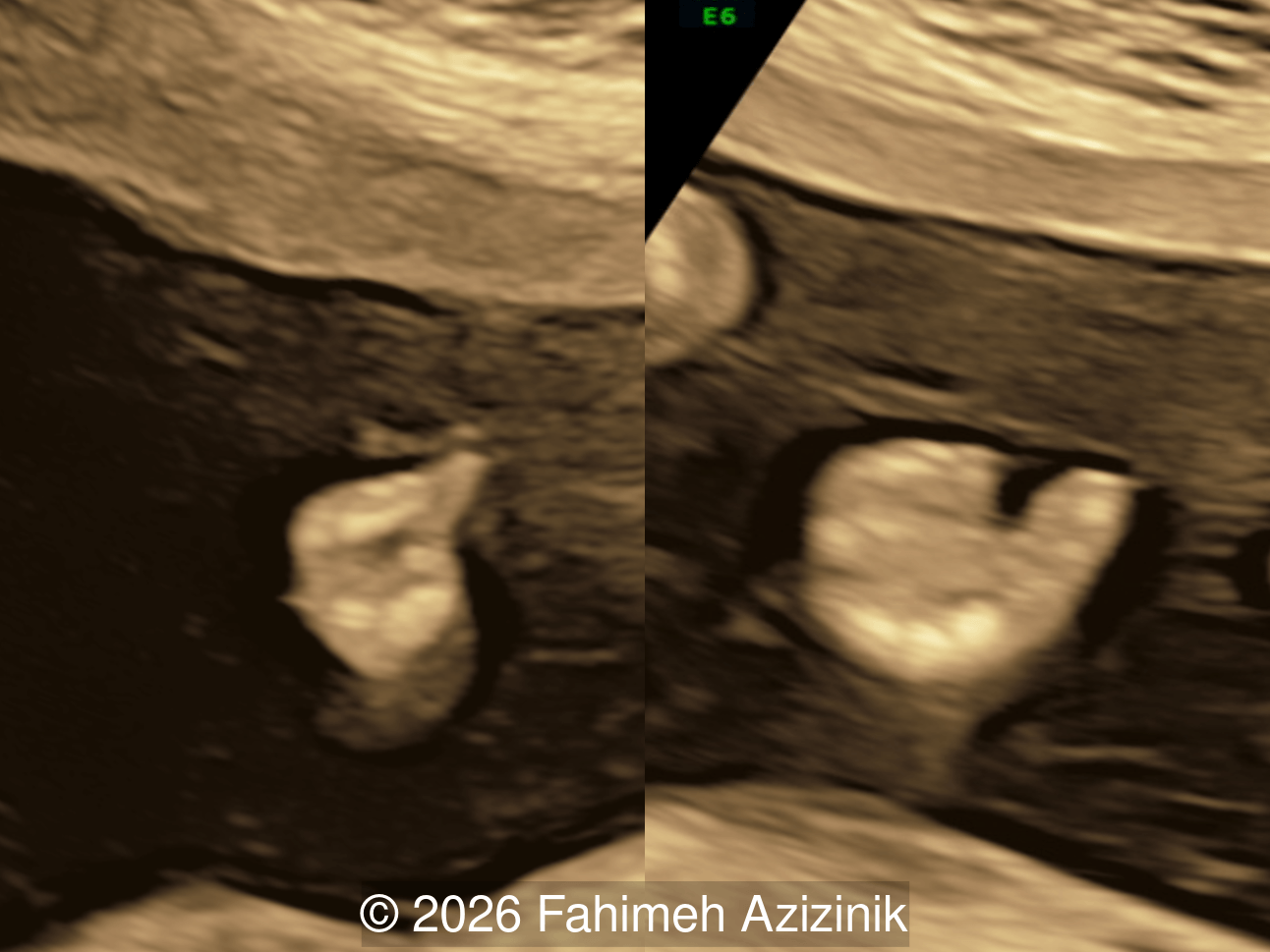
- Complex syndactyly of the hands and feet. Complex hand brachysyndactyly with “mitten-like” appearance due to bony fusion of the fingers
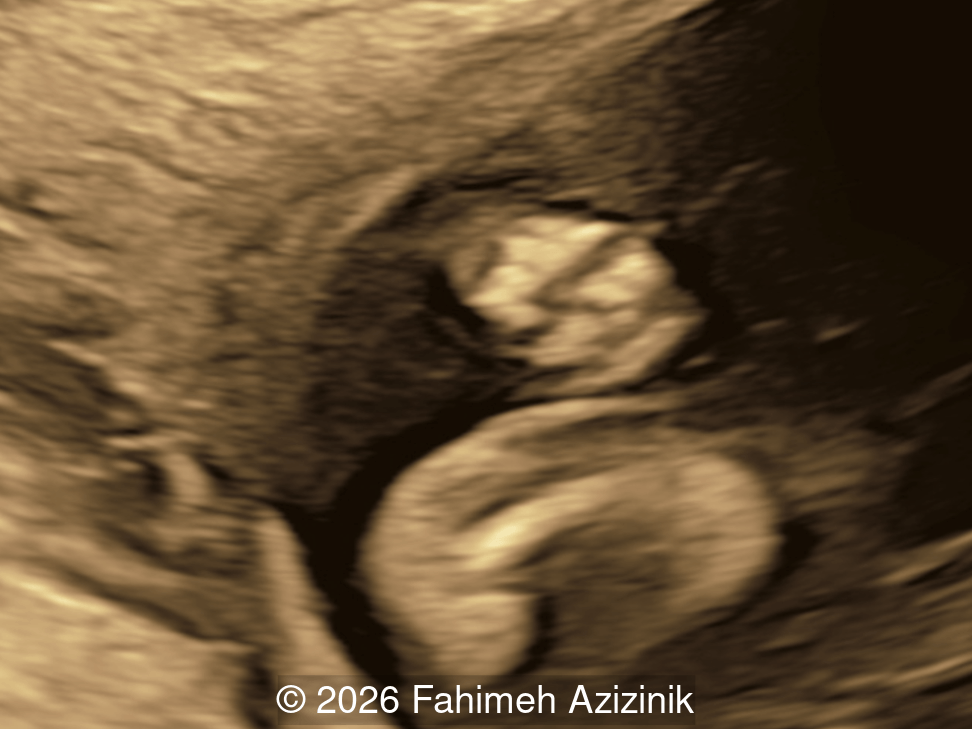
- Bicoronal craniosynostosis
- Hypertelorism
- Frontal bossing
Apert syndrome was confirmed by molecular testing. The parents decided to terminate the pregnancy.
Discussion
Acrocephalosyndactyly syndromes are a group of congenital malformation disorders characterized by craniofacial anomalies resulting from premature craniosynostosis and variable degrees of syndactyly of the fingers and/or toes [1,2]. They have classically been divided into five subtypes: I (Apert syndrome), II (Apert-Crouzon syndrome), III (Saethre-Chotzen syndrome), IV (Goodman syndrome), and V (Pfeiffer syndrome). Historically, the classification of these syndromes has been inconsistent with initially proposed clinical categories discarded or redefined, and other presentations previously considered acrocephalosyndactylies reclassified to acrocephalopolysyndactylies, which is characterized by the additional presence of polydactyly. Considerable phenotypic overlap occurs, so investigators now consider many of these syndromes as variants of the same diseases.
Craniosynostosis is defined as the premature fusion of one or more cranial sutures, which leads to secondary distortion of skull shape due to restricted growth perpendicular to the fused suture and compensatory overgrowth at the non-fused sutures [3,4]. Single‑sutural craniosynostosis is typically an isolated, non‑syndromic finding. Nevertheless, a subset of cases, particularly those involving the coronal sutures, may have an underlying genetic etiology. Conversely, in syndromic craniosynostosis mutations are common, most frequently involving the fibroblast growth factor receptor genes (FGFR2 and FGFR3), as well as TWIST1 and EFNB1 [5]. FGFR-related craniosynostosis syndromes are characterized by bicoronal craniosynostosis, distinctive facial features, and variable hand and foot findings. Prenatal diagnosis of isolated cases of craniosynostosis is infrequent as the characteristic findings on ultrasound, which include decreased hypoechoic space between the skull bones, altered cranial shape, abnormal cephalic index, and the "brain shadowing sign", are not evident during the routine mid-trimester scan [6,7]. These syndromes share characteristic craniofacial features, often referred to as a ‘Crouzenoid face’, including midfacial retrusion, hypertelorism, proptosis due to shallow orbits, a high‑arched palate, flattened malar region, and a beaked nose [3,8].
Apert syndrome (AS, OMIM # 101200), also known as acrocephalosyndactyly type 1 and Apert-Crouzon disease, is characterized by the triad of abnormal cranial shape, midface hypoplasia and bilateral syndactyly of the hands and feet [9]. There is an increased risk of mild-to-moderate intellectual disability, although cognitive impairment is not universal [10]. Although it was first reported by Wheaton in 1894 [11], it takes its name from the French pediatrician Eugène Apert [12], who published a series of nine cases in 1906 that presented a triad of craniosynostosis, syndactyly and maxillary hypoplasia.
Birth prevalence of the Apert syndrome is estimated to be approximately 1 in 65,000 to 80,000 newborns and accounts for about 4.5% of all cases of craniosynostosis [13,14]. Apert syndrome is an autosomal dominant disease, though most cases are the result of de novo mutations of FGFR2 gene located on chromosome 10q26. Studies show that 98% of the cases result from one of two missense mutations in the FGFR2 gene (S252W and P253R) [15]. The S252W mutation is the most common, occurring in 63% of patients and associated with more severe craniofacial anomalies, whereas the P253R may be associated with more severe syndactyly [16]. These mutations usually have a paternal origin and are associated with advanced paternal age, in part due to the increased frequency of mutations in sperm [14,17-18].
Bicoronal synostosis is the characteristic cranial hallmark of Apert syndrome, detectable sonographically from 18 weeks. Its most recognizable manifestation is turribrachycephaly, defined by an elevated cephalic index (typically >85–90%) and a tall cranial vault with marked frontal bossing [19]. On prenatal ultrasound, the face in Apert syndrome shows midface hypoplasia with a depressed nasal bridge, proptosis, hypertelorism, and frontal bossing. The extremities demonstrate syndactyly of both bone and soft tissue of the hands and feet with partial-to-complete fusion often involving second, third, and fourth digits, known as “mitten hands” and “socked feet”. In severe cases, all digits are fused, with the presence of a single nail known as “synonychia” [20]. Other ultrasound findings include mild ventriculomegaly, agenesis of the corpus callosum, deficient or absent septum pellucidum, and fusion of the cervical vertebrae at the level of C5-C6 [21]. Cardiovascular (atrial and ventricular septal defect) and genitourinary anomalies (hydronephrosis, and cryptorchidia) are present in 10% of patients [22]. Increased nuchal translucency in the first trimester [23], widely open metopic suture [24], and polyhydramnios [25] have also been reported. Three-dimensional (3D) ultrasound facilitates assessment of surface abnormalities of the face and extremities, and magnetic resonance imaging (MRI) can be used to evaluate associated intracranial conditions [26].
Following an initial ultrasound suspicion of Apert syndrome, a detailed and targeted ultrasound (preferably using 3D/4D modalities) should be performed to confirm and characterize the extent of the abnormalities, followed by magnetic resonance imaging (MRI) to rule out associated structural brain abnormalities and evaluate the cortical maturation. The definitive diagnosis is obtained after an invasive procedure (amniocentesis) for targeted sequencing of the FGFR2 gene and confirmation of the molecular diagnosis [19].
The differential diagnosis of Apert syndrome includes other acrocephalosyndactyly syndromes, including Crouzon, Pfeiffer, Carpenter and Saethre-Chotzen, in which the pattern of syndactyly constitutes a fundamental discriminating criterion [27,28]. The absence of limb abnormalities, in conjunction with milder craniosynostosis, is indicative of Crouzon syndrome. The craniosynostosis and exophthalmos observed in Pfeiffer syndrome are more severe than those typically found in Apert syndrome. Additionally, Pfeiffer syndrome presents with broad abducted thumbs, broad great toes, and brachymesophalangy and partial syndactyly of the hands and feet. Carpenter syndrome typically presents with polydactyly, whereas Saethre‑Chotzen syndrome may show a partial duplication of the hallux, in addition to mild syndactyly, as the most characteristic distal limb anomalies.
References
- Prevel CD, Eppley BL, McCarty M. Acrocephalosyndactyly syndromes: a review. J Craniofac Surg. 1997 Jul;8(4):279-285.
- Acrocephalosyndactyly Syndromes. In: Syndromes: Rapid Recognition and Perioperative Implications. Bissonnette B, ed. McGraw-Hill Companies, Inc., New York, NY, USA, 2006; pg 11-18.
- Johnson D, Wilkie AO. Craniosynostosis. Eur J Hum Genet. 2011 Apr;19(4):369-376.
- Ketwaroo PD, Robson CD, Estroff JA. Prenatal Imaging of Craniosynostosis Syndromes. Semin Ultrasound CT MR. 2015 Dec;36(6):453-464.
- Roscioli T, Elakis G, Cox TC, et al. Genotype and clinical care correlations in craniosynostosis: findings from a cohort of 630 Australian and New Zealand patients. Am J Med Genet C Semin Med Genet. 2013 Nov;163C(4):259-270.
- Massoud M, Bault J-P, and Cabet S. Antenatal Diagnosis of Craniosynostosis. In: Di Rocco F and Kestle J, ed. Neurosurgical Aspects of Craniosynostosis. Springer Nature Switzerland AG, 2024; pg 253-267.
- Krajden Haratz K, Leibovitz Z, et al. The 'Brain Shadowing Sign': A Novel Marker of Fetal Craniosynostosis. Fetal Diagn Ther. 2016;40(4):277-284.
- Heuzé Y, Martínez-Abadías N, Stella JM, et al. Quantification of facial skeletal shape variation in fibroblast growth factor receptor-related craniosynostosis syndromes. Birth Defects Res A Clin Mol Teratol. 2014 Apr;100(4):250-259.
- OMIM. APERT SYNDROME [Internet]. Baltimore (MD): Johns Hopkins University; c1966– [updated 2022 Jan 25; cited 2026 Jan 31]. Available from: https://www.omim.org/entry/101200.
- David DJ, Anderson P, Flapper W, et al. Apert Syndrome: Outcomes From the Australian Craniofaciafromit's Birth to Maturity Management Protocol. J Craniofac Surg. 2016 Jul;27(5):1125-1134.
- Wheaton SW: Two specimens of congenital cranial deformity in infants associated with fusion of the fingers and toes. Trans Pathol Soc London. 1894; 45:238-241.
- Apert E. De l’acrocéphalosyndactylie. Bull Mém Soc Med Hop Paris. 1906; 23: 1310-1313.
- Cohen MM Jr, Kreiborg S, Lammer EJ, et al. Birth prevalence study of the Apert syndrome. Am J Med Genet. 1992 Mar 1;42(5):655-659.
- Tolarova MM, Harris JA, Ordway DE, Vargervik K. Birth prevalence, mutation rate, sex ratio, parents' age, and ethnicity in Apert syndrome. Am J Med Genet. 1997 Nov 12;72(4):394-398.
- Wilkie AO, Slaney SF, Oldridge M, et al. Apert syndrome results from localized mutations of FGFR2 and is allelic with Crouzon syndrome. Nat Genet. 1995 Feb;9(2):165-172.
- von Gernet S, Golla A, Ehrenfels Y, et al. Genotype-phenotype analysis in Apert syndrome suggests opposite effects of the two recurrent mutations on syndactyly and outcome of craniofacial surgery. Clin Genet. 2000 Feb;57(2):137-139.
- Erickson JD, Cohen MM Jr. A study of parental age effects on the occurrence of fresh mutations for the Apert syndrome. Ann Hum Genet. 1974 Jul;38(1):89-96.
- Glaser RL, Broman KW, Schulman RL, et al. The paternal-age effect in Apert syndrome is due, in part, to the increased frequency of mutations in sperm. Am J Hum Genet. 2003 Oct;73(4):939-947.
- Li H, Shen J, Tang M, et al. From FGFR2 mutations to precision management: a review of prenatal diagnosis and multidisciplinary interventions in apert syndrome. Front Pediatr. 2025 Nov 5;13:1658654.
- Apert Syndrome. In: Atlas of Genetic Diagnosis and Counseling, 3rd edition. Chen, H, ed. Springer Science+Business Media LLC, New York, NY, USA, 2017; pg 151-169.
- Breik O, Mahindu A, Moore MH, et al. Central nervous system and cervical spine abnormalities in Apert syndrome. Childs Nerv Syst. 2016 May;32(5):833-838.
- Cohen MM Jr, Kreiborg S. Visceral anomalies in the Apert syndrome. Am J Med Genet. 1993 Mar 15;45(6):758-760.
- Aleem S, Howarth ES. Apert syndrome associated with increased fetal nuchal translucency. Prenat Diagn. 2005 Nov;25(11):1066-1067.
- Faro C, Chaoui R, Wegrzyn P, et al. Metopic suture in fetuses with Apert syndrome at 22-27 weeks of gestation. Ultrasound Obstet Gynecol. 2006 Jan;27(1):28-33.
- Chen CP, Lin SP, Su YN, et al. Apert syndrome associated with upper airway obstruction and gastroesophageal reflux inducing polyhydramnios in the third trimester. Taiwan J Obstet Gynecol. 2010 Jun;49(2):231-234.
- Werner H, Castro P, Daltro P, et al. Prenatal diagnosis of Apert syndrome using ultrasound, magnetic resonance imaging, and three-dimensional virtual/physical models: three case series and literature review. Childs Nerv Syst. 2018 Aug;34(8):1563-1571.
- Chen CP, Su YN, Hsu CY, et al. Second-trimester molecular prenatal diagnosis of sporadic Apert syndrome following sonographic findings of mild ventriculomegaly and clenched hands mimicking trisomy 18. Taiwan J Obstet Gynecol. 2010 Mar;49(1):129-132.
- Kumari K, Saleh I, Taslim S, et al. Unraveling the Complexity of Apert Syndrome: Genetics, Clinical Insights, and Future Frontiers. Cureus. 2023 Oct 18;15(10):e47281.
Discussion Board
Winners

Paula Melone United States Physician

Azar Farajov Azerbaijan Physician

Yulia Sologub Russian Federation Physician

Pawel Swietlicki Poland Physician

belen garrido Spain Physician

Andrii Averianov Ukraine Physician

Alexandr Krasnov Ukraine Physician

Mayank Chowdhury India Physician

Nutan Thakur India Physician

Vladimir Lemaire United States Physician

Boujemaa Oueslati Tunisia Physician

Tatiana Koipish Belarus Physician

carlos lopez Venezuela Physician

CHARLES SARGOUNAME India Physician

Bahauddin Sallout Saudi Arabia Physician

Yuliya Taustukha Belarus Physician

tina walden United States Sonographer

Amparo Gimeno Spain Physician

Elena Andreeva Russian Federation Physician

CRISTINA MARTINEZ PAYO Spain Physician

ALBANA CEREKJA Italy Physician

Eti Zetounie Israel Sonographer

Eda Özden Tokalıoğlu Turkey Physician

SAMUEL GELVEZ TELLEZ Colombia Physician

Murat Cagan Turkey Physician

Sonio Sonio France AI
Mária Brešťanská Slovakia Physician
Nguyen Thac Viet Viet Nam Physician

Büşra Cambaztepe Turkey Physician
Gayane Begjanyan Armenia Physician
Đặng Mai Quỳnh Viet Nam Physician

Hien Nguyen Van Viet Nam Physician

Almaz Kinzyabulatov Russian Federation Physician

Kareem Haloub Australia Physician

Dr Monika Sharma India Physician

Anette Beverdam Netherlands Sonographer

Fred Pop Uganda Sonographer

Annette Reuss Germany Physician

Arati Appinabhavi India Physician

Seadet Zeynalova Azerbaijan Physician

shruti Agarwal India Physician

Vu The Anh Viet Nam Physician

YULIA VISHNEVSKAYA Russian Federation Physician

Sruthi Pydi India Physician

Oanh Oanh Viet Nam Physician

philip pattyn Belgium Physician

Nguyễn Lê Hoàng Viet Nam Physician

Denys Saitarly Israel Physician

Le Tien Dung Viet Nam Physician

Tetiana Ishchenko Ukraine Physician

Costin Radu Lucian Romania Physician

Le Duc Viet Nam Physician

Mậu Nguyễn Bá Viet Nam Physician

Annet Esveld Netherlands Sonographer

Philippe Viossat Antarctica Consultant

Dr Abhinaya Subramaniam India Physician

Gnanasekar Periyasamy India Physician

Petra Tallova Slovakia Physician

ZHANNA Kurmangaliyeva Kazakhstan Physician

Murad Esetov Russian Federation Physician

jimena salcedo Spain Physician

Carmie Cee Australia Physician

Maria Bulanova Russian Federation Physician

Simen Vergote Canada Physician

Mert Eyupoglu Turkey Physician

Gulten Rafibeyli Azerbaijan Physician

Ayten Sadigova Azerbaijan Physician

Gulsum Mammadova Azerbaijan Physician

Aynur Garibova Azerbaijan Physician

Ulviyya Jafarova Azerbaijan Physician

Aida Nadirova Azerbaijan Physician

Elnara Baghirova Azerbaijan Physician

Elnara Suleymanova Azerbaijan Physician

Mikael Huhtala Finland Physician

Nataliia Antonenko Ukraine Physician

Smurova lidia Russian Federation Physician

Balgis Suliman United States Sonographer

Rajkumar Pachlaniya India Physician

inshirah sgayer Israel Sonographer