Case of the Week #633
(1) Femicare, Center of prenatal ultrasonographic diagnostics, Martin, Slovakia; (2) UCSF Health, San Francisco, California, United States
23-year-old primigravid woman with non-contributive medical history, came to our office at 19 weeks, 5 days gestation for routine second trimester screening. The following observations were seen:
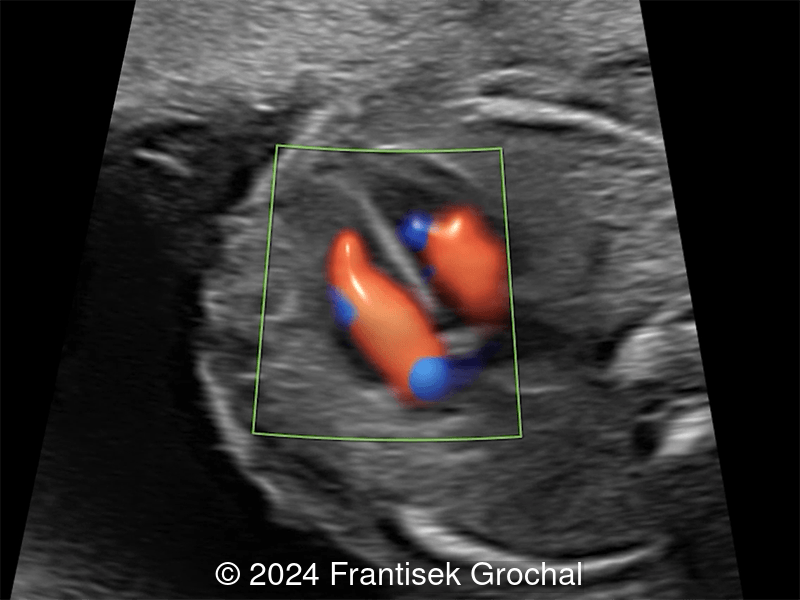
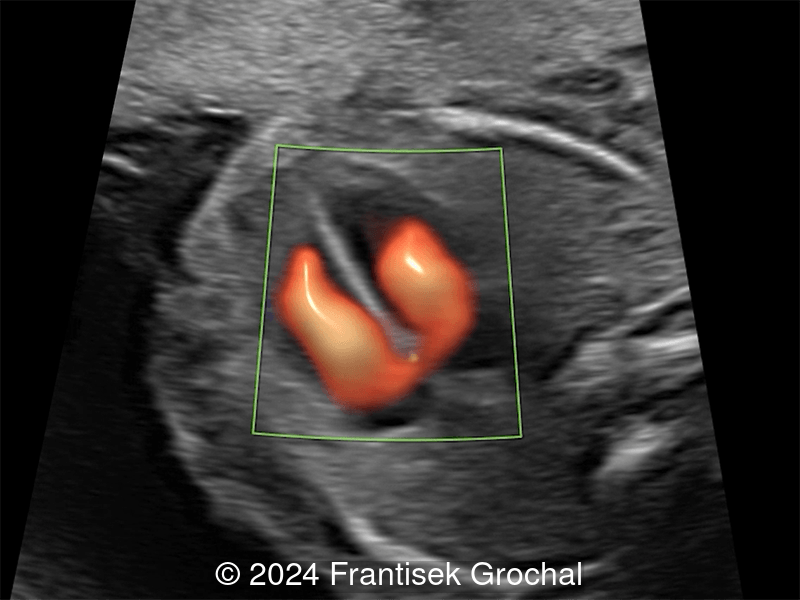
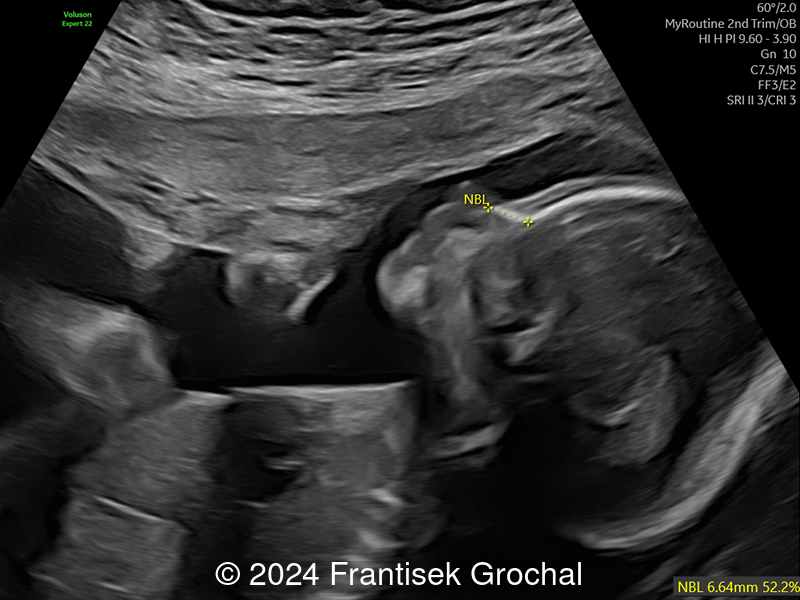
View the Answer Hide the Answer
Answer
We present a case of sternal defect, also known as sternal cleft or sternoschisis.
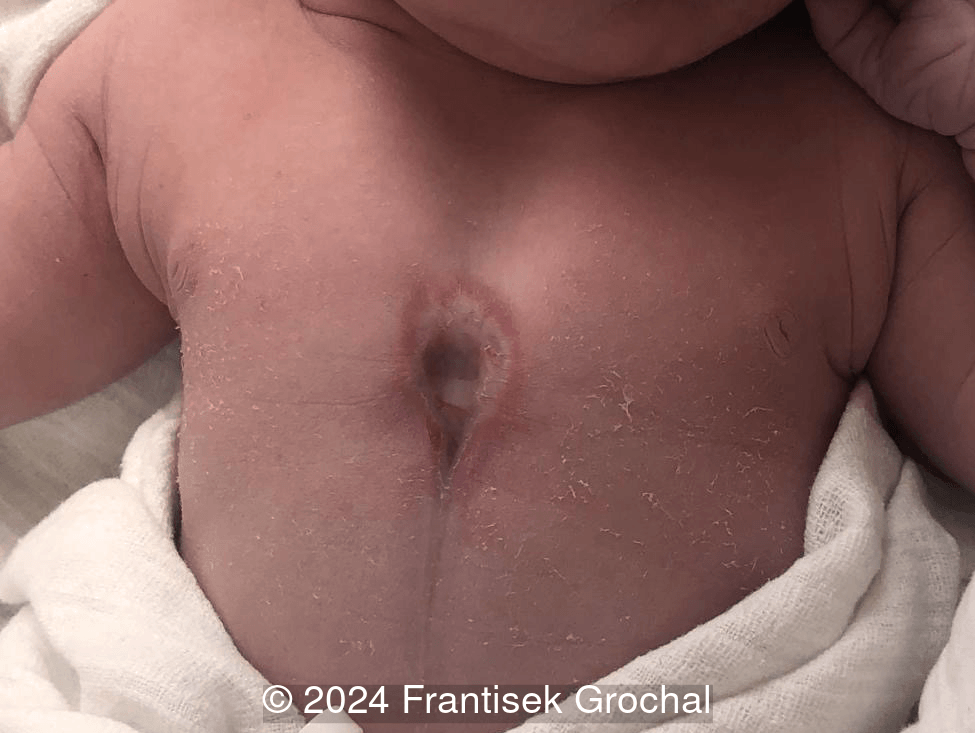
Images reveal a normal heart with a thinned midline chest wall.
The neonate was born at term. On exam, there was a midline raphe, though no cutaneous hemangiomas. Eye exam was normal. Ultrasound of the sternum confirmed a V-shaped defect of the upper sternum. Postnatal echocardiography demonstrated an atrial septal defect. The patient underwent surgical correction at one month of age.
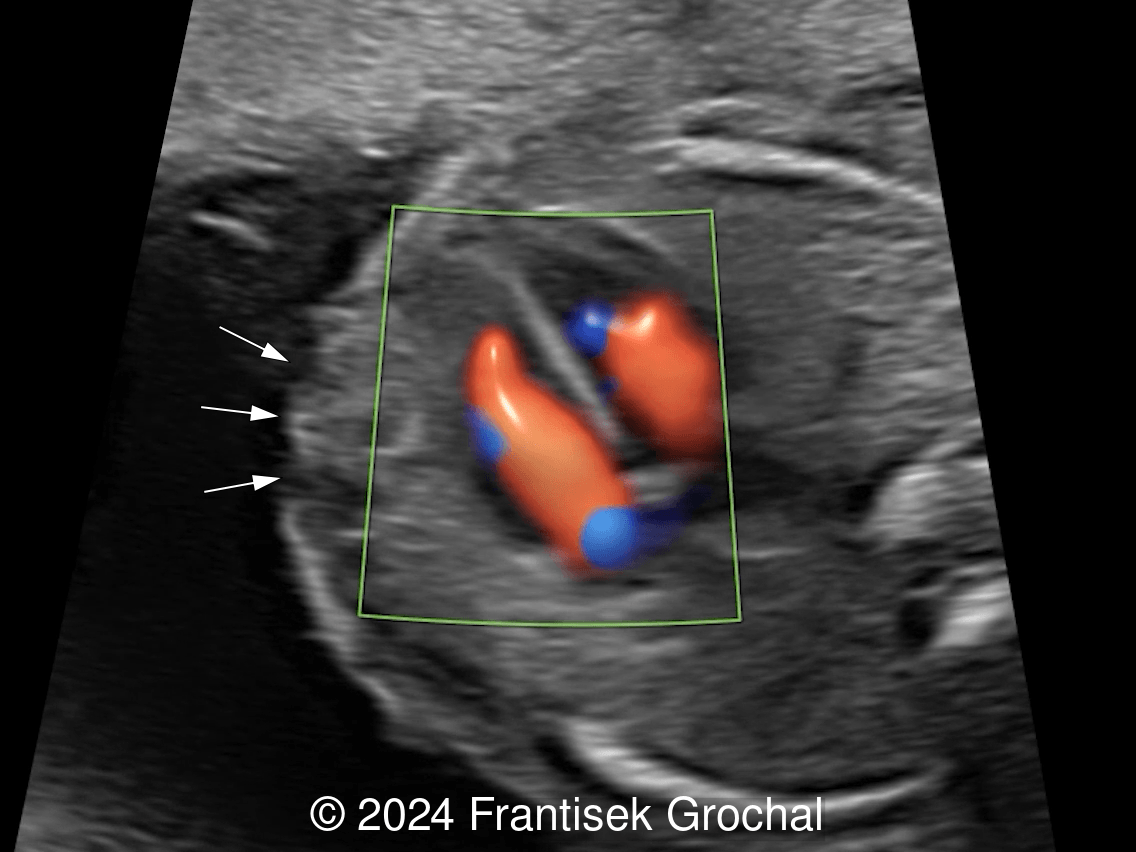
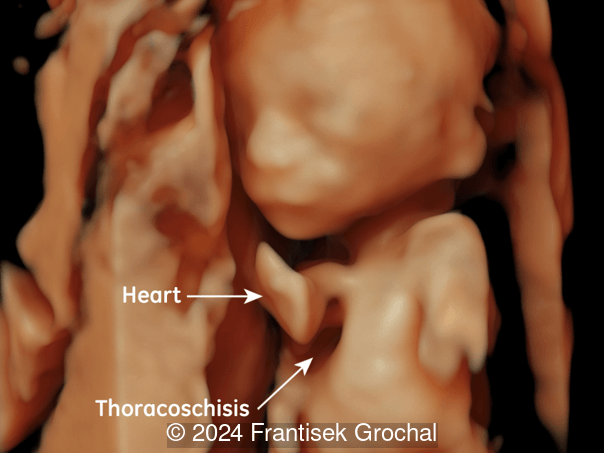
The spectrum of findings may differ in severity with inconspicuous variants as demonstrated in this case. In contrast, a sternal defect associated with ectopia cordis is usually striking and easily recognized during prenatal ultrasonography.
Discussion
A sternal defect occurs when there is separation of the sternum in the midline chest. This may or may not occur with ectopia cordis. One of the first reported cases of a sternal defect with ectopic cordis was in 1740 by de Torres in which he documents the observations of the physician to the King of Spain who describes a male infant with a heart that "burst through the breast, and having broke the sternum, appeared on the outside" [1].
The first anatomic classification of sternal defects was proposed by Weese in 1818 who defined three types: ectopia cordis with sternal cleft, ectopia suprathoracic, and ectopia subthoracic [2]. Several classifications were described over the following century and a half [3], and in 1962 Kanagasuntheram et al suggested five categories of ectopia cordis and associated sternal defects: 1) cervical, in which the heart is in the cervical region with a sternum that is usually intact; 2) thoraco-cervical, in which the heart is partially in the cervical region but the upper portion of the sternum is defective; 3) thoracic, in which the sternum is defective and the heart lies partially or completely outside the thorax; 4) thoraco-abdominal, in which the ectopic position of the heart is accompanied by a cleft sternum, a diaphragmatic defect and a midline abdominal defect allowing the heart to enter the abdomen and usually referred to as Cantrell's syndrome; and 5) abdominal, in which a diaphragmatic defect allows the heart to enter the abdomen [4]. Leca et al reviewed over 200 cases of ectopic cordis in the literature and found that cervical and thoraco-cervical types occur in 9% of cases, thoracic in 37%, thoracoabdominal in 36% and abdominal in 11% [5]. In 1990, Shamberger and Welch reviewed a case series of 16 sternal defects at Boston Children’s hospital and added sternal cleft or bifid sternum as a category. These cases are described as having an orthotopic heart, an intact pericardium, and normal skin coverage [6]. The incidence of sternal cleft is not known, though in a study reviewing over 5000 patients with chest wall malformations between 1987 and 2001, 8 (0.15%) had a sternal cleft [7]. Acastello et al suggested a classification of sternal clefts into partial, located in the upper or lower sternum, or complete with involvement the entire sternum [7]. In a study reviewing 115 sternal clefts, 65% were the superior partial type, 27% were the complete type, and 8% were the inferior partial type [8]. Females are more commonly affected with sternal clefts [7,9].
The sternum has three parts: the manubrium, the mesosternum composed of four segments, and the metasternum or xiphoid process [10]. Development of the sternum appears during the 6th week as a pair of parallel mesenchymal bands. Cells migrate from the lateral plate mesoderm on either side of the anterior chest wall to fuse in the midline by the 10th week of gestation. The sternal bands join at their cephalic ends and gradually fuse in the midline, progressing caudally [11]. Mice studies demonstrate that the sternal elements arise in situ, and the medial movement occurs in the absence of ribs [12]. Ossification initially starts in the manubrium and upper part of the sternal body at the 6th month of development, the middle of the body in the 7th month, and the lower body in the first postnatal year. Coalescence of these ossification centers does not take place until after puberty [10,13].
It is difficult to conceive of one pathogenic mechanism that could account for all types of ectopia cordis and associated sternal defects. A popular theory suggests that amniotic bands may play a role in mechanical teratogenesis. Rupture of the chorion or yolk sac at 3 weeks of gestation may result in interference of the normal descent of the heart. This may explain thoracic ectopia cordis with cephalic pointing of the cardiac apex and thoracoabdominal ectopia cordis in which the heart is tethered to paraumbilical structures by bands [14,15]. Other studies suggest genetic mutations may contribute to the pathogenesis of sternal defects. The bone morphogenic protein (BMP) pathway is involved in the ventral folding of the embryo. Mutations in BMP can result in defective folding and contribute to ectopic cordis [16] or sternal cleft [17]. Homeobox-containing genes are involved in segmental differentiation, specifying positional information along the anterior-posterior body axis. Mice with defective HoxB expression present with abnormalities of sternal fusion [18].
A sternal defect could also be considered a thoracoschisis as the etymology of the word “thoraco” meaning "of or pertaining to the thorax,” and “schisis” from Greek meaning "division or cleft. ” The term “thoracoschisis” was first used by in 1977 by pediatric surgeons in South Africa to describe an infant born with a left intercostal defect containing herniated liver, colon, stomach and omentum, as well as left diaphragmatic hernia and ipsilateral reduction deformity of the upper limb [19]. Thoracoschisis has therefore traditionally been used to describe a rare condition in which there is a thoracic defect with evisceration of abdominal organs often with associated diaphragmatic hernia and limb anomalies [20,21]. Fewer than 20 cases have been reported in the literature [21-23]. Some authors have considered it as part of the limb body wall complex (LBWC) spectrum [20,24]. The pathogenesis of LBWC is unknown however several theories exist. Early amniotic sac rupture can result in formation of amniotic bands which entrap and disrupt limb formation [25]. However, amniotic bands do not easily explain the internal anomalies found in LBWC [26]. Alternatively, Van Allen et al suggested that a vascular disruption explains both the internal malformations and the limb defects. Embryonic blood supply at 4-6 weeks gestation is divided into quadrants with the right and left aorta supplying the embryo and the right and left anterior and posterior cardinal veins draining the embryo. Damage to one of these vessels could lead to regional developmental disruption as is seen in LBWC [26]. Finally, genetic mutations resulting in abnormal embryonic folding have been implicated in LBWC and include genes in the Frizzled-PCP signaling and Wnt-PCP pathway (Ptk7, Vangl2, Scribble, and Ryk), as well as genes involved in the Homeobox pathway (Pitx2, Msx1/Msx2) [27]. Interestingly, many of the disrupted pathways causing LBWC are also involved in the phenotype of split sternum (mutations in Hoxb2/Hoxb4 genes involved in Homeobox pathway; mutations in Wnt5a genes involved in Wnt signaling) and ectopic cordis (mutations in Wls, Lrp5/6 receptors, and Porcupine genes involved in Wnt signaling) [27].
In patients with sternal defects, approximately 40-50% have associated anomalies [7,8], with the upper partial sternal defect being the type that is most commonly isolated (55%) [8]. Cardiac defects have been found in 22-25% of patients [7-9]. Cervicofacial hemangiomas are found in 10-13% [7,8,28] and can be found in association with PHACES syndrome which describes Posterior fossa brain malformations (cerebellum hypoplasia, cortical dysgenesis, arachnoidal cysts), capillary Hemangiomas, Arterial malformations (stenosis or agenesis, especially in the carotid and vertebral arteries), Cardiac malformations and aortic coarctation, Eye abnormalities (microphtalmos, colloboma, optics nerve hypoplasia), and Sternal defects [29,30]. Cervicofacial hemangiomas are more commonly associated with upper partial sternal defects and complete sternal defects, while lower partial defects are more commonly associated with pentalogy of Cantrell [8]. Approximately 8% of patients with sternal defects have a recognized syndrome, including PHACES and pentalogy of Cantrell [8].
Prenatal diagnosis of sternal cleft is difficult. In normal pregnancies, the sternum appears as ‘a chain of beads’ extending from the manubrium downwards, and is first visualized at 19 weeks, at which time two to three ossification centers are demonstrated. The fourth ossification center appears at approximately 22 weeks, and the fifth center at 29 weeks [31]. In fetuses with sternal cleft, the midline anterior thoracic wall may be thin and depressed, and seen to transmit cardiac pulsations [30]. The sternal cleft can result in paradoxical motion of the chest with fetal respiratory motion [32] or hiccupping [33]. MRI can be used as adjunct in diagnosis [30,34]. Only 9 cases of sternal defect have been described in the literature [30,32-39]. Of these cases (Table 1), 6 were partial upper sternal clefts, 2 were complete clefts and 1 was a partial lower cleft. Cardiac defects were found in 2 cases and included an ascending aortic aneurysm and ventricular septal defect. Venous malformations were found in 3 cases. The earliest diagnosis of sternal cleft was at 20 weeks gestation [37].
Study | GA (weeks) | Sex | Type | Cardiac anomalies | Venous anaomalies | Other anomalies | Prenatal Imaging | Outcome |
|---|---|---|---|---|---|---|---|---|
GA: gestational age; US: ultrasound; MRI: magnetic resonance imaging; SVC: superior vena cava; TOP: termination of pregnancy; VSD: ventricular septal defect; NA: not available. | ||||||||
Mahone 1992 | 32 | F | partial, upper | none | none | none | US shows absent midline sternal calcification | surgery |
Rose 1996 | 22 | F | partial, upper | none | none | chest wall hamartoma | US, MRI showed lobulated chest wall mass, incomplete visualization of the sternum | surgery |
Twomey 2005 | 24 | F | complete | none | bilateral SVC with the left SVC draining into dilated coronary sinus | none | US shows thin and sunken midline anterior chest wall; paradoxical motion with respiration | surgery |
Thebault 2009 | 3rd trimester | F | partial, upper | none | cutaneous hemangiomas; intra-phrenic draining of renal veins, duplication of SVC which flows into right atrium | supraumbilical raphe | US, MRI shows thinned and sunken midline anterior chest wall | no surgery |
Izquierdo 2009 | 20 | F | complete | none | none | midline raphe, abnormal insertion of sternocleidomastoid muscles | US shows absence of manubrium, wide separation of rib cage, transmission of heart impulse to overlying skin | TOP |
Pasoglou 2012 | 32 | NA | partial, lower | VSD | none | supraumbilical raphe | US, MRI shows thin midline thoracic wall with an underlying pulsating heart | surgery |
Yuksel 2016 | NA | F | partial, upper | none | congenital venous malformation | rhabdomyoma of larynx | NA | surgery |
Lu 2022 | 21 | NA | partial, upper | ascending aortic aneurysm | none | supraumbilical raphe | US shows wide separation of the clavicular heads | TOP |
Perdomo 2024 | 26 | M | partial, upper | none | none | partial agenesis of the right clavicle | US, MRI shows thin thoracic wall, deep depression in middle of chest when fetus hiccupped | surgery |
Surgical repair of sternal defects is recommended as the paradoxical motion of the chest wall with respiration can lead to bony destruction of the thorax, impaired cardiac or pulmonary function, and respiratory tract infection [9]. Repair within the first months of life is ideal as the chest wall is pliable and primary closure can often be achieved. In older children, surgical techniques for closure of the sternal defect become more complex and may require chondrotomies, osteotomies, or clavicular dislocation to increase the chest wall compliance, thymectomy to reduce mediastinal compression, and muscle flaps, bone grafts, or prosthetic material for closure of the sternal defect [8,9].
References
[1] De Torres JI. Extract of a letter from Jos. Ignat. de Torres, M. D. to the Royal Society, containing an extraordinary case of the heart of a child turned upside down. Philosophical Transactions of the Royal Society of London. 1741 Dec;41(461):776-778.
[2] Weese, Carl. De cordis ectopia. Dissertatio inauguralis anatomico‑pathologica, Berlin: J. F. Stark, 1818.
[3] Gabriel A, Donnelly J, Kuc A, et al. Ectopia cordis: a rare congenital anomaly. Clin Anat. 2014 Nov;27(8):1193-9.
[4] Kanagasuntheram R, Verzin JA. Ectopia cordis in man. Thorax. 1962 Jun;17(2):159-67.
[5] Leca F, Thibert M, Khoury W, et al. Extrathoracic heart (ectopia cordis). Report of two cases and review of the literature. Int J Cardiol. 1989 Feb;22(2):221-8.
[6] Shamberger RC, Welch KJ. Sternal defects. Pediatr Surg Int. 1990 May;5:156–164.
[7] Acastello E, Majluf R, Garrido P, et al. Sternal cleft: a surgical opportunity. J Pediatr Surg. 2003 Feb;38(2):178-83.
[8] Hinchcliff KM, Xue Y, Wong GB. Reconstruction of Congenital Sternal Cleft: A Systematic Review of the Literature. Ann Plast Surg. 2021 May 1;86(5S Suppl 3):S418-S421.
[9] Torre M, Rapuzzi G, Carlucci M, et al. Phenotypic spectrum and management of sternal cleft: literature review and presentation of a new series. Eur J Cardiothorac Surg. 2012 Jan;41(1):4-9.
[10] O'Neal ML, Dwornik JJ, Ganey TM, et al. Postnatal development of the human sternum. J Pediatr Orthop. 1998 May-Jun;18(3):398-405.
[11] Mekonen HK, Hikspoors JPJM, Mommen G, et al. Development of the ventral body wall in the human embryo. J Anat. 2015 Nov;227(5):673-85.
[12] Chen JM. Studies on the morphogenesis of the mouse sternum. II. Experiments on the origin of the sternum and its capacity for self-differentiation in vitro. J Anat. 1952 Oct;86(4):387-401.
[13] Engum SA. Embryology, sternal clefts, ectopia cordis, and Cantrell’s pentalogy. Semin Pediatr Surg. 2008 Aug;17(3):154-60.
[14] Haynor DR, Shuman WP, Brewer DK, et al. Imaging of fetal ectopia cordis: roles of sonography and computed tomography. J Ultrasound Med. 1984 Jan;3(1):25-7.
[15] Van Allen MI, Myhre S. Ectopia cordis thoracalis with craniofacial defects resulting from early amnion rupture. Teratology. 1985 Aug;32(1):19-24.
[16] Gavrilov S, Lacy E. Genetic dissection of ventral folding morphogenesis in mouse: embryonic visceral endoderm-supplied BMP2 positions head and heart. Curr Opin Genet Dev. 2013 Aug;23(4):461-9.
[17] Goldman DC, Donley N, Christian JL. Genetic interaction between Bmp2 and Bmp4 reveals shared functions during multiple aspects of mouse organogenesis. Mech Dev. 2008 Dec 11;126(3-4):117–127.
[18] Ramírez-Solis R, Zheng H, Whiting J, et al. Hoxb-4 (Hox-2.6) mutant mice show homeotic transformation of a cervical vertebra and defects in the closure of the sternal rudiments. Cell. 1993 Apr 23;73(2):279-94.
[19] Davies MR, Rode H, Cywes S. "Thoracoschisis" associated with an ipsilateral distal phocomelia and an anterolateral diaphragmatic hernia-a case report. J Pediatr Surg. 1977 Oct;12(5):755-7.
[20] Hanafi HR, Zakaria ZA. Prenatal Diagnosis of Thoracoschisis and Review of Literature. Case Rep Obstet Gynecol. 2017 Nov 16;2017:9821213.
[21] Ragan M, Mathur K, Hartwich J. A novel surgical approach to thoracoschisis. A novel surgical approach to thoracoschisis. J Pediatr Surg Case Rep. 2021 Oct;73:102014.
[22] Ardıçlı B, Karaman A, Özyazıcı A. et al. Isolated thoracoschisis: Case report. Turk J Pediatr 2017; 59(2): 217-220.
[23] Abdelkader BS, Abdelbary MAA, Ahmed AN. Isolated thoracoschisis. J Pediatr Surg Case Rep. 2020 Jun;60:101538.
[24] Vujovic D, Sretenovic A, Raicevic M, et al. Thoracoschisis associated with Limb Body Wall Complex. APSP J Case Rep. 2017 May 1;8(3):19.
[25] Miller ME, Graham Jr JM, Higginbottom MC, et al. Compression-related defects from early amnion rupture: evidence for mechanical teratogenesis. J Pediatr. 1981 Feb;98(2):292-7.
[26] Van Allen MI, Curry C, Gallagher L. Limb body wall complex: 1, Pathogenesis. Am J Med Genet. 1987 Nov;28(3):529-48.
[27] Formstone C, Aldeiri B, Davenport M, et al. Ventral body wall closure: Mechanistic insights from mouse models and translation to human pathology. Dev Dyn. 2025 Feb;254(2):102-141.
[28] Hersh JH, Waterfill D, Rutledge J, et al. Sternal Malformation / Vascular Dysplasia Association. Am J Med Genet. 1985 May;21(1):177-86, 201-2.
[29] James PA, McGaughran J. Complete Overlap of PHACE Syndrome and Sternal Malformation—Vascular Dysplasia Association. Am J Med Genet. 2002 Jun 1;110(1):78-84.
[30] Thébault N, Le Guern H, Le Fiblec B, et al. Prenatal diagnosis of a complete sternal cleft in a child with PHACES syndrome—a case report. Prenat Diagn. 2009 Feb;29(2):179-81.
[31] Zalel Y, Lipitz S, Soriano D, et al. The development of the fetal sternum: a cross-sectional sonographic study. Ultrasound Obstet Gynecol. 1999 Mar;13(3):187-90.
[32] Twomey EL, Moore AM, Ein S, et al. Prenatal ultrasonography and neonatal imaging of complete cleft sternum: a case report. Ultrasound Obstet Gynecol. 2005 Jun;25(6):599-601.
[33] Perdomo L, Caminal J, Rodríguez M, et al. EP06.03: Prenatal diagnosis of sternal cleft. Ultrasound Obstet Gynecol. 2024 Sep;64(Suppl.1):149.
[34] Pasoglou V, Tebache M, Rausin L, et al. Sternal cleft: Prenatal multimodality imaging. Pediatr Radiol. 2012 Aug;42(8):1014-6.
[35] Mahone PR, Sherer DM, D'Angio C, et al Prenatal ultrasonographic findings associated with wide clefting of the upper two thirds of the fetal sternum. Am J Obstet Gynecol. 1992 Apr;166(4):1219-21.
[36] Rose NC, Coleman BG, Wallace D, et al. Prenatal diagnosis of a chest wall hamartoma and sternal cleft. Ultrasound Obstet Gynecol. 1996 Jun;7(6):453-5.
[37] Izquierdo MT, Bahamonde A, Domene J. Prenatal Diagnosis of a Complete Cleft Sternum With 3-Dimensional Sonography. J Ultrasound Med. 2009 Mar;28(3):379-83.
[38] Yuksel M, Kuru P, Ermerak NO, et al. Intrauterine diagnosed sternal cleft patient and her management. J Vis Surg. 2016 Mar 14;2:48.
[39] Lu J, Tse WT, Law KM, et al. Prenatal diagnosis of ascending aortic aneurysm associated with sternal cleft. Ultrasound Obstet Gynecol. 2022 Feb;59(2):267-268.
A special thanks to Dr. Roberto Romero and Dr. Javier Cortejoso for their expert input regarding this case.