Case of the Week # 590
(1) SDMH Hospital Jaipur; (2) Centro Médico Recoletas, Valladolid, Spain
30-year-old woman, G2P1, with no significant obstetric, medical and family history, presented at 13 weeks 4 days for nuchal translucency screen. The following images were obtained.
View the Answer Hide the Answer
Answer
We present a case of arthrogryposis multiplex congenita.
The images revealed:
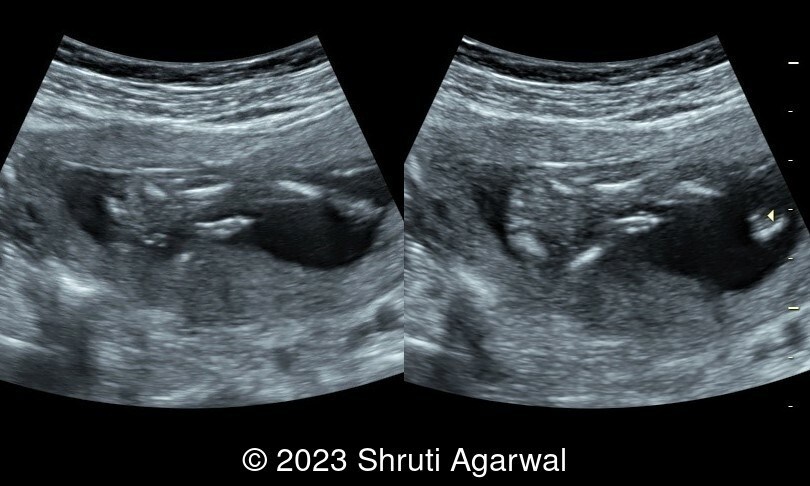
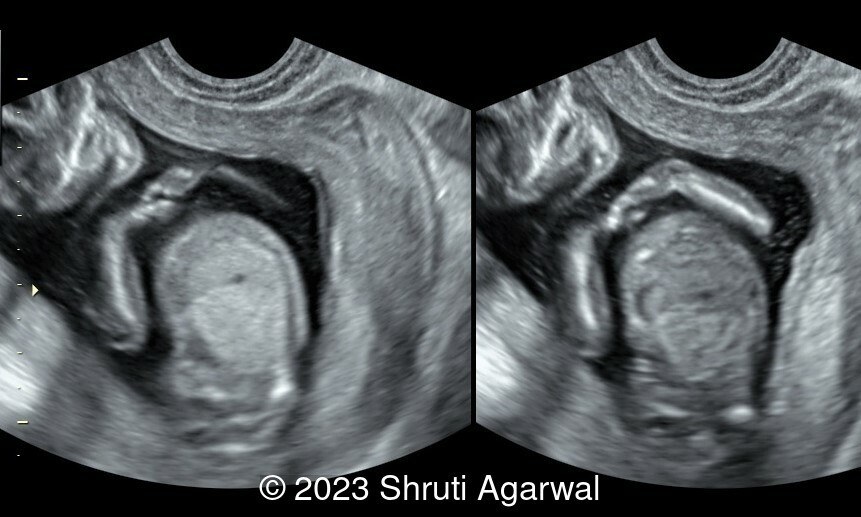
- Persistent hyperextended lower limb, flexed at the hip and knee joint (Image 1). Club foot was identified on the other limb (Image 2).
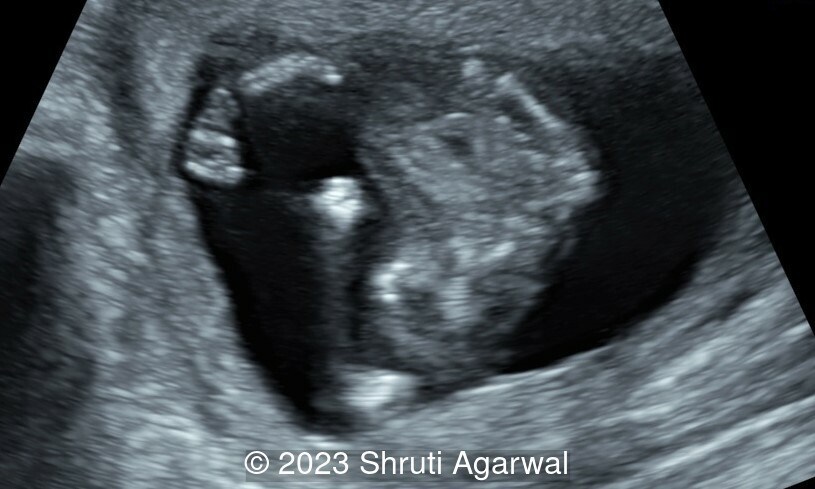
- Both upper limbs were flexed in front of the chest (Image 3).
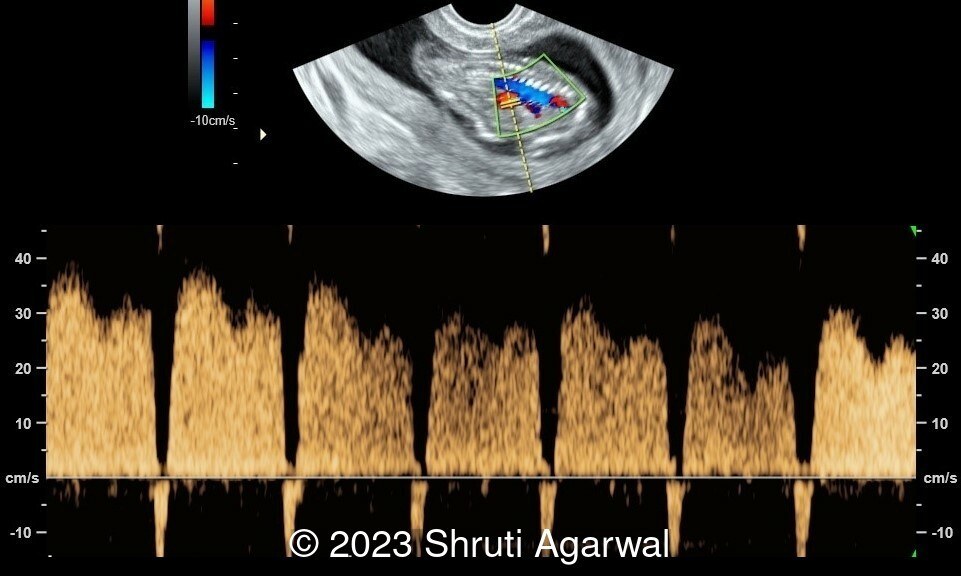
- Reversal of flow in the ductus venosus (Image 4).
- Persistently abnormal position of fetus with very limited movement of torso (Video 1).
Repeat ultrasound scan at 24 hours demonstrated the fetus in the same position.
Discussion
Congenital contractures are present in 1 in every 100-200 newborns and can be divided into two categories: isolated congenital contractures (clubfoot or dislocated hips), and syndromes that present with multiple congenital contractures [1]. The terms arthrogryposis and arthrogryposis multiplex congenita include a large group of syndromes characterized by two or more congenital joint contractures affecting different areas of the body [2, 3]. It is a sign rather than a diagnosis caused by lack of fetal movement, and can result from multiple pathologic mechanisms. Lack of fetal movement leads to excess deposition of connective tissue around the joints and shortening of tendons, which decreases mobility and leads to joint contractures. It is also known as “fetal akinesia deformation sequence.”
The term “arthrogryposis” is derived from Greek, meaning “curved and hooked joints”. It was described for the first time by Otto in 1841 [4], and subsequently termed “multiple congenital contractures” by Schantz in 1897 [5], and “arthrogryposis” by Rosenkranz [6]. The name “arthrogryposis multiplex congenita” was coined in 1923 by Stern in a report of four cases of congenital multiple ankylosis involving the joints of the extremities [7]. The overall prevalence of arthrogryposis is estimated to be about 1 in 3,000 to 1 in 5,000 live births [8]. Prenatal prevalence is likely higher as some cases are associated with fetal demise. Arthrogryposis multiplex congenita includes more than 400 different disorders, which include brain malformations, neuromuscular diseases, connective tissue disorders, and chondrodysplasias, which are often of genetic origin [1, 3].
Postnatally, three categories of congenital contractures are distinguished based on clinical findings [9]. Each one represents approximately a third of the total:
- primarily limb involvement
- musculoskeletal involvement plus other system anomalies
- musculoskeletal involvement plus lethality, central nervous system dysfunction, and/or significant mental retardation.
In prenatal life, it is more useful to distinguish between intrinsic fetal conditions versus extrinsic and environmental factors that could be contributing to the limb contractures [2, 10, 11]. Extrinsic conditions that limit the intrauterine space are multiple births (triplets, quadruplets, etc), decreased amniotic fluid, and rarely uterine malformation or large uterine fibroids. Maternal conditions such as myasthenia gravis, diabetes mellitus, multiple sclerosis, and myotonic dystrophy, exposure to medication (muscle relaxants, some antiepileptics, misoprostol), recreational drugs (cocaine), and viral infections (rubella, varicella, and Zika virus) have been reported to be associated with fetal akinesia [12]. Intrinsic fetal conditions include functional abnormalities of the brain, spinal cord, peripheral nerves, neuromuscular junction, muscles, bones, tendons, joints, and restrictive dermopathies [11]. The likelihood of finding an underlying chromosomal abnormality (trisomy 18, mosaic trisomy 8, and microdeletions including contiguous gene deletion syndromes) or genetic disease is increased if the multiple congenital contractures are associated with fetal structural anomalies [1]. Arthrogryposis may be inherited as an autosomal recessive, autosomal dominant, or X-linked trait, in addition to some cases of maternal inheritance due to mitochondrial disorders [1, 12]. Currently, more than 400 genes have been reported to have mutations associated with arthrogryposis [13]. In addition to the karyotype and a chromosome microarray, it is important to carry out a single gene testing in families with a known aetiology, gene panel testing, or whole exome/genome sequencing [1, 11, 12].
Common causes of arthrogryposis of fetal/intrinsic origin are amyoplasia and distal arthrogryposis. Amyoplasia (the most common form of arthrogryposis encompassing about one third of all cases) appears sporadically, possibly being of vascular and/or hypoxic origin in the first or early second trimester [14]. A genetic etiology is unlikely, given that amyoplasia has been described in one twin of a spontaneous monozygotic pregnancy while the other was normal [15].
The ultrasound diagnosis is based on the lack of movement of the extremities and the persistent malposition of the limbs [11]. The absence of movements is verified if they are not observed during a 30–45-minute scan (given that 13 minutes is the maximal pause between consecutive movements in a normal population at 20 weeks gestational age) [16]. First-trimester ultrasound findings can include increased nuchal translucency and cystic hygroma. Indirect ultrasound signs of reduced fetal movement are polyhydramnios and collapsed stomach which may be a result of impaired swallowing, and facial anomalies such as micro-/retrognathia [1, 11, 12]. When extrinsic conditions seem unlikely, and the contracture pattern does not point to amyoplasia (elbows extended, wrists flexed, hands clenched, shoulders internally rotated, knees fixed in extension or flexion and feet in equinovarus position), the cause of multiple contractures is most likely genetic [1]. Since detailed evaluation of fetal movements is not routinely performed during prenatal ultrasound, approximately 75% of cases are not diagnosed before birth [17].
There is no in utero treatment for arthrogryposis. Fetal immobility and disuse of limbs can lead to reduced bone growth, hypomineralization and fractures of the long bones, persistence of breech position and need for delivery by caesarean section to avoid fractures [11, 14]. The prognosis of Fetal Akinesia Dysmorphic Sequence will be conditioned by its cause, the number and severity of contractures, and the need for ventilation at birth, which confers a worse prognosis. If parents choose to interrupt the pregnancy or a neonatal death occurs, the family should be offered an autopsy and genetic studies to try to identify the cause of arthrogryposis [11]. In cases where a specific cause is not identified, the risk of recurrence in subsequent pregnancies is 3% to 5% or 7% in cases of central nervous system involvement [2].
References:
[1] Filges I, Jünemann S, Viehweger E, Tercanli S. Fetal arthrogryposis-what do we tell the prospective parents? Prenat Diagn. 2023 Jun;43(6):798-805. doi: 10.1002/pd.6299. Epub 2023 Jan 12. PMID: 36588183.
[2] Rink BD. Arthrogryposis: a review and approach to prenatal diagnosis. Obstet Gynecol Surv. 2011 Jun;66(6):369-77. doi: 10.1097/OGX.0b013e31822bf5bb. PMID: 21851751.
[3] Filges I, Tercanli S, Hall JG. Fetal arthrogryposis: Challenges and perspectives for prenatal detection and management. Am J Med Genet C Semin Med Genet. 2019 Sep;181(3):327-336. doi: 10.1002/ajmg.c.31723. Epub 2019 Jul 18. PMID: 31318155.
[4] The classic. A human monster with inwardly curved extremities. By Adolph Wilhelm Otto, 1841. Clin Orthop Relat Res. 1985 Apr;(194):4-5. PMID: 3884208.
[5] Schantz A. Ein fall von multiplen kongenitalen kontrakturen. Z Orthop Chir 1898; 5: 9. 12.
[6] Rosenkranz E. Ueber kongenitale kontrakturen der oberen extremitaten. Z Orthop Chir 1905; 14: 52. 13.
[7] Stern WG. Arthrogryposis multiplex congenita. JAMA. 1923;81(18):1507–1510. doi:10.1001/jama.1923.02650180025009
[8] Lowry RB, Sibbald B, Bedard T, Hall JG. Prevalence of multiple congenital contractures including arthrogryposis multiplex congenita in Alberta, Canada, and a strategy for classification and coding. Birth Defects Res A Clin Mol Teratol. 2010 Dec;88(12):1057-61. doi: 10.1002/bdra.20738. Epub 2010 Nov 15. PMID: 21157886.
[9] Hall JG. Arthrogryposes (multiple congenital contractures). In: Rimoin DL, Pyeritz RE, Korf BR, editors. Emery and Rimoin’s principle and practice of medical genetics, sixth ed. Churchill Livingstone, New York, 2013; Chapter 161, pp. 1-101.
[10] Hall JG. Arthrogryposis (multiple congenital contractures): diagnostic approach to etiology, classification, genetics, and general principles. Eur J Med Genet. 2014 Aug;57(8):464-72. doi: 10.1016/j.ejmg.2014.03.008. Epub 2014 Apr 3. PMID: 24704792.
[11] Niles KM, Blaser S, Shannon P, Chitayat D. Fetal arthrogryposis multiplex congenita/fetal akinesia deformation sequence (FADS)-Aetiology, diagnosis, and management. Prenat Diagn. 2019 Aug;39(9):720-731. doi: 10.1002/pd.5505. Epub 2019 Jul 16. PMID: 31218730.
[12] Skaria P, Dahl A, Ahmed A. Arthrogryposis multiplex congenita in utero: radiologic and pathologic findings. J Matern Fetal Neonatal Med. 2019 Feb;32(3):502-511. doi: 10.1080/14767058.2017.1381683. Epub 2017 Sep 27. PMID: 28954562.
[13] Kiefer J, Hall JG. Gene ontology analysis of arthrogryposis (multiple congenital contractures). Am J Med Genet C Semin Med Genet. 2019 Sep;181(3):310-326. doi: 10.1002/ajmg.c.31733. Epub 2019 Aug 1. PMID: 31369690.
[14] Hall JG, Aldinger KA, Tanaka KI. Amyoplasia revisited. Am J Med Genet A. 2014 Mar;164A(3):700-30. doi: 10.1002/ajmg.a.36395. Epub 2014 Jan 23. PMID: 24459070.
[15] Hall JG. The mystery of monozygotic twinning II: What can monozygotic twinning tell us about Amyoplasia from a review of the various mechanisms and types of monozygotic twinning? Am J Med Genet A. 2021 Jun;185(6):1822-1835. doi: 10.1002/ajmg.a.62177. Epub 2021 Mar 25. PMID: 33765349.
[16] de Vries JI, Fong BF. Normal fetal motility: an overview. Ultrasound Obstet Gynecol. 2006 Jun;27(6):701-11. doi: 10.1002/uog.2740. PMID: 16710877.
[17] Filges I, Hall JG. Failure to identify antenatal multiple congenital contractures and fetal akinesia--proposal of guidelines to improve diagnosis. Prenat Diagn. 2013 Jan;33(1):61-74. doi: 10.1002/pd.4011. PMID: 23296716.
Discussion Board
Winners

Dianna Heidinger United States Sonographer

Javier Cortejoso Spain Physician

Igor Yarchuk United States Sonographer

Iuliia Iudina Russian Federation Physician

Ana Ferrero Spain Physician

Mayank Chowdhury India Physician

Boujemaa Oueslati Tunisia Physician

Halil Mesut Turkey Physician

Anita Silber Israel Physician

Kimberly Delaney United States Sonographer
Marianovella Narcisi Italy Physician

Shari Morgan United States Sonographer

ALBANA CEREKJA Italy Physician

Eslam Adel ammar Egypt Physician

Rasha Abo Almagd Egypt Physician

Ionut Valcea Romania Physician

Cong Bang Duong Viet Nam Physician

Hien Nguyen Van Viet Nam Physician

Borisova Elena Russian Federation Physician

CHERYL TURNER United States Sonographer

SAVITA SHIRODKAR India Physician

Andrea Stoop-Berends Netherlands Sonographer

Sruthi Pydi India Physician

cara gavagni United States Sonographer