Case of the Week #568
(1) Imagerie Medicale du Capricorne, Reunion Island; (2) Obstetrics department, Felix Guyon Hospital, Reunion Island; (3) Clinical genetics department, Felix Guyon Hospital, Reunion Island; (4) Neonatalogy department, Felix Guyon Hospital, Reunion Island
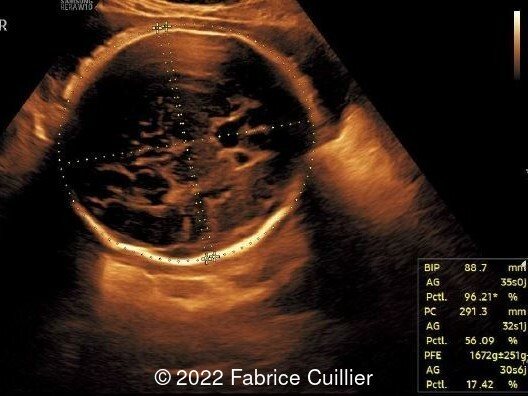
Case Report: A 20-year-old primigravida with no significant past medical or family history presented for evaluation. First-trimester ultrasound was normal, and combined screening was low-risk. During her 2nd trimester ultrasound, micropenis and unilateral foot malposition were noted. There was normal appearance of the heart, thorax, abdomen, stomach, bowel, kidneys, bladder, spine, and upper limbs (not shown). Fetal growth was normal. The following images were obtained during the 3rd trimester ultrasound at 31 weeks.
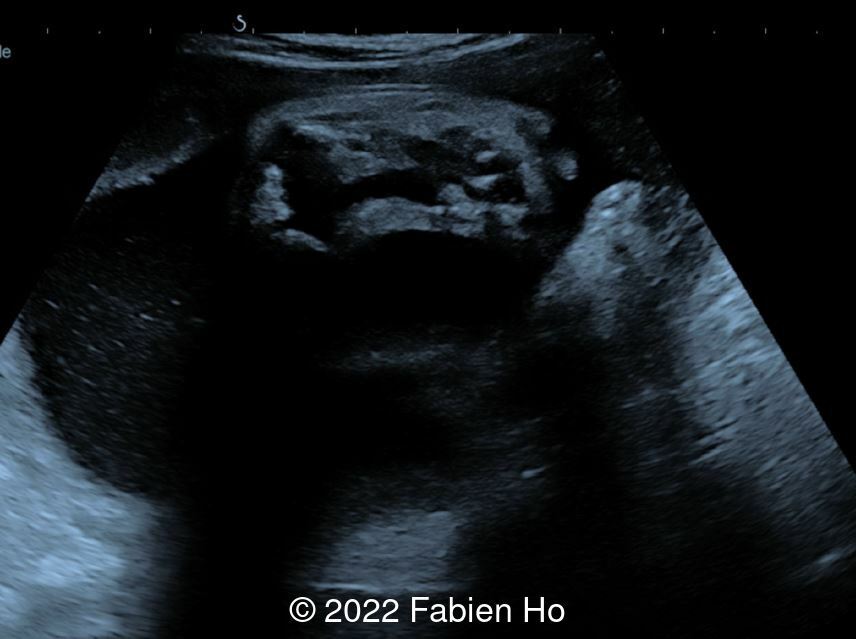
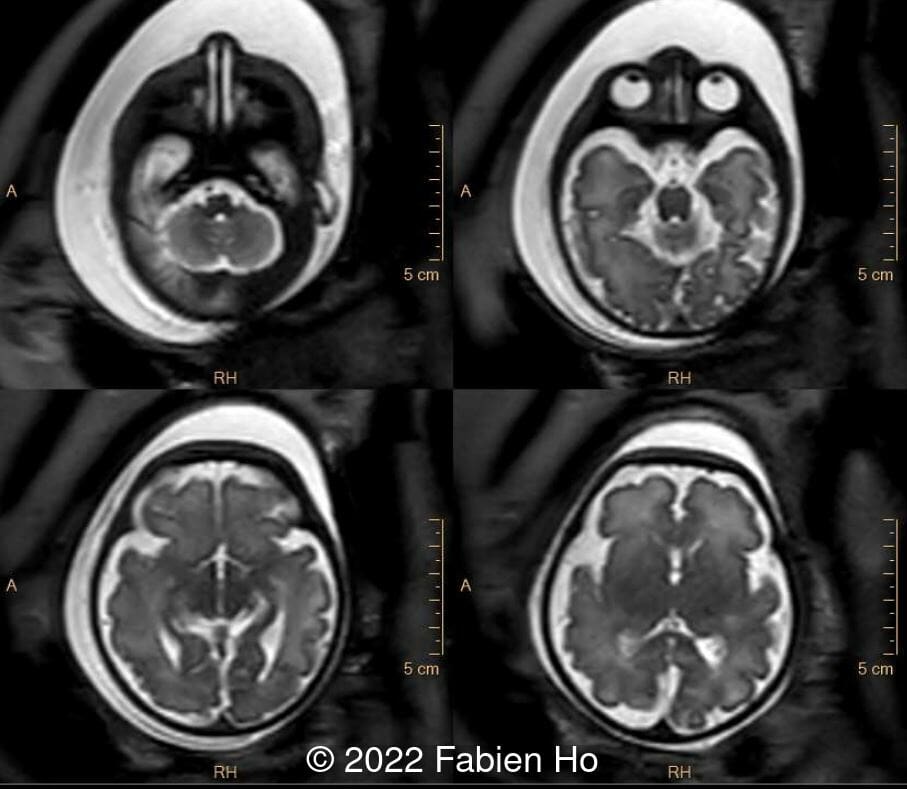
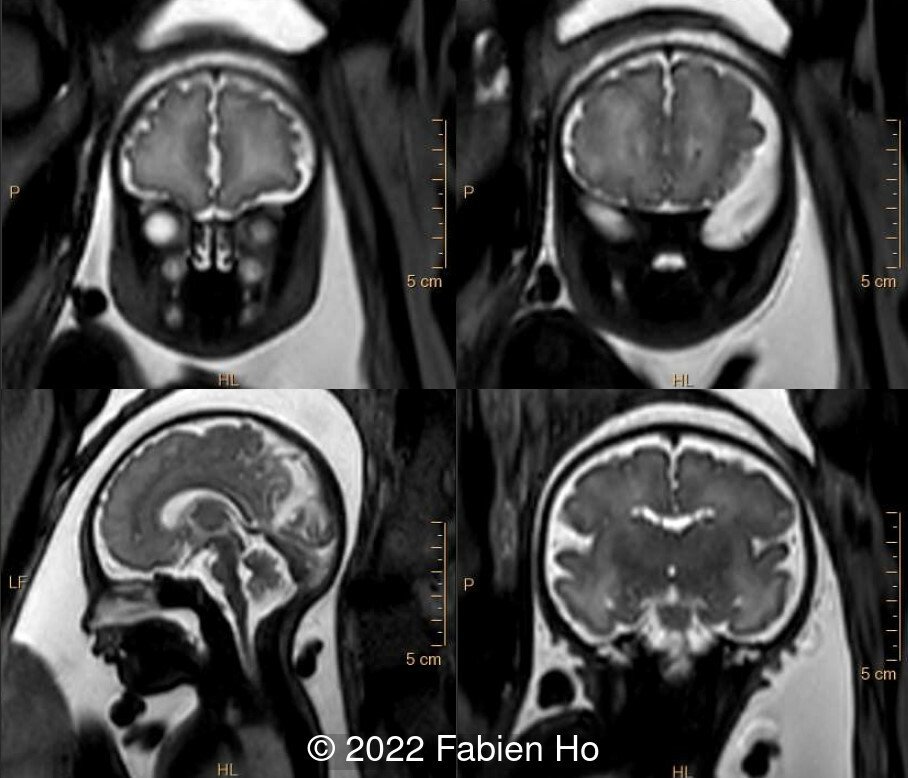
Fetal brain MRI at 33 weeks gestation showed the following:
Amniocentesis was performed. Karyotype and microarray were normal. An amniotic fluid 7-dehydrocholesterol level (testing for Smith-Lemli-Optiz Syndrome) was normal. Whole exome sequencing was offered but declined at 34 weeks of gestation.
View the Answer Hide the Answer
Answer
We present a case of CHARGE syndrome (OMIM #214800).
- Image 1: Brachycephaly with normal neuroanatomy
- Image 2: Normal appearance of the orbits and eyes
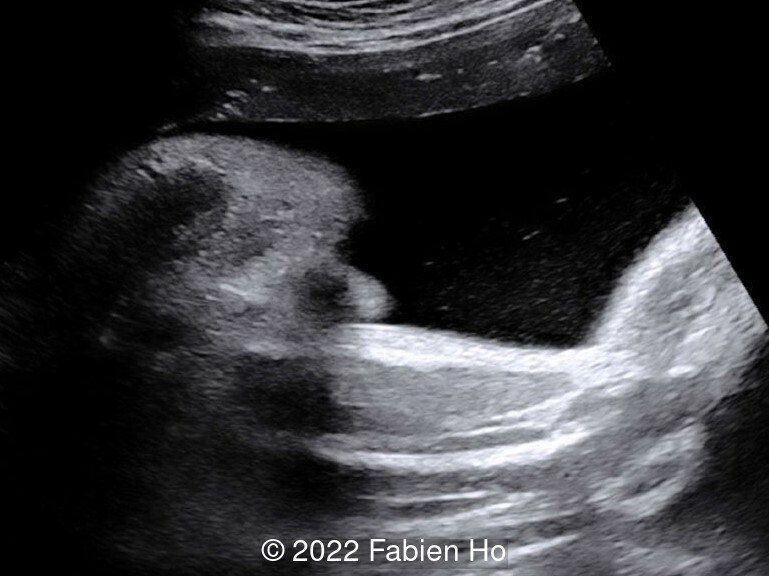
- Image 3: Normal appearance of fetal nose and lips
- Image 4: Fetal hard palate demonstrating no cleft.
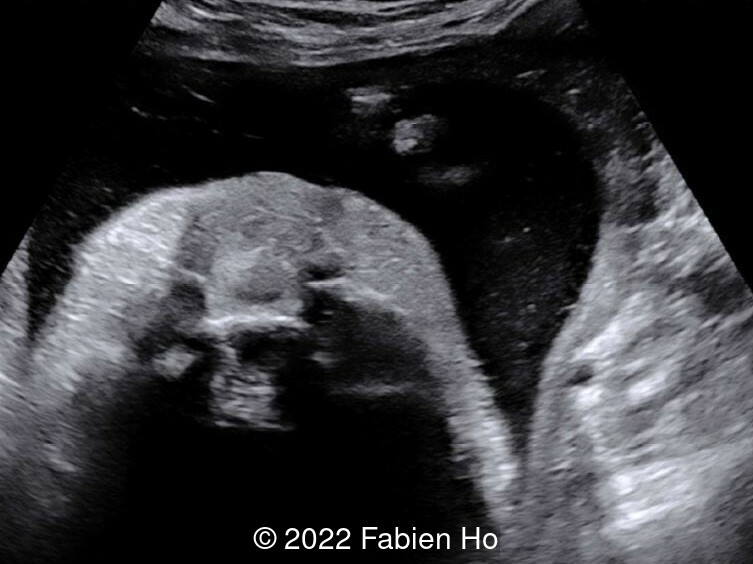
- Image 5: Normal facial profile
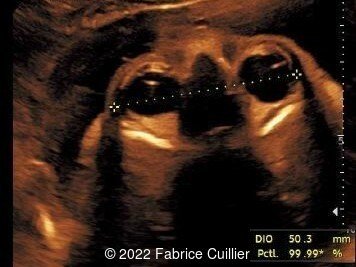
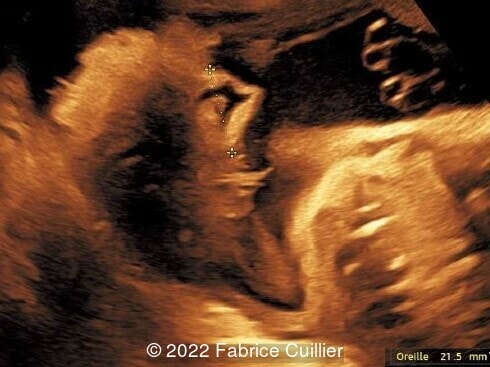
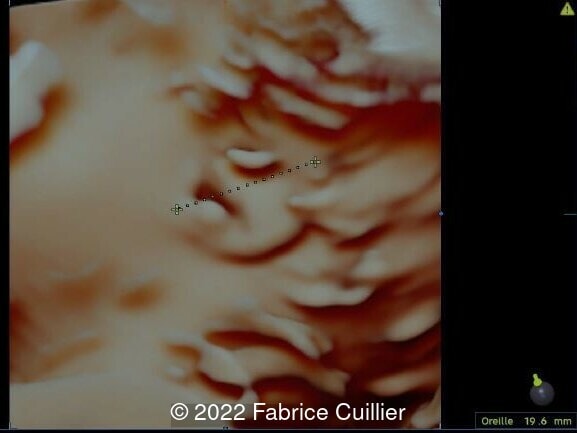
- Image 6 and Image 7: Abnormal, square-shaped ear
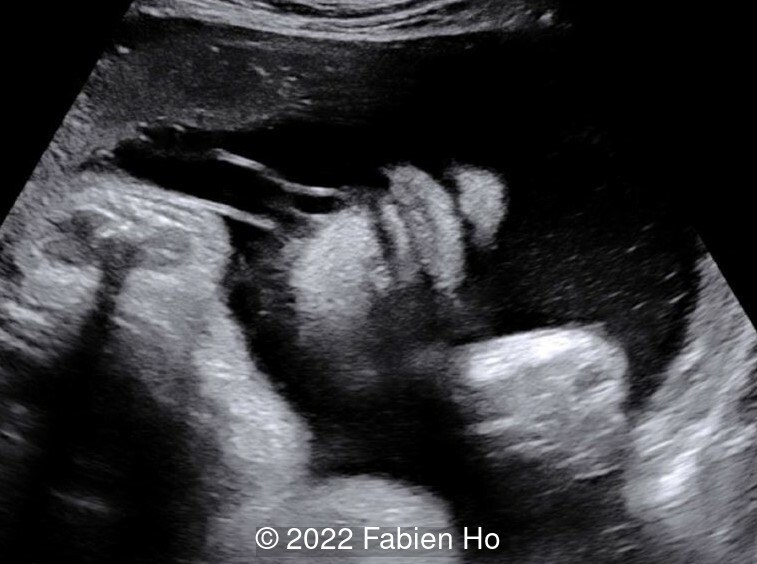
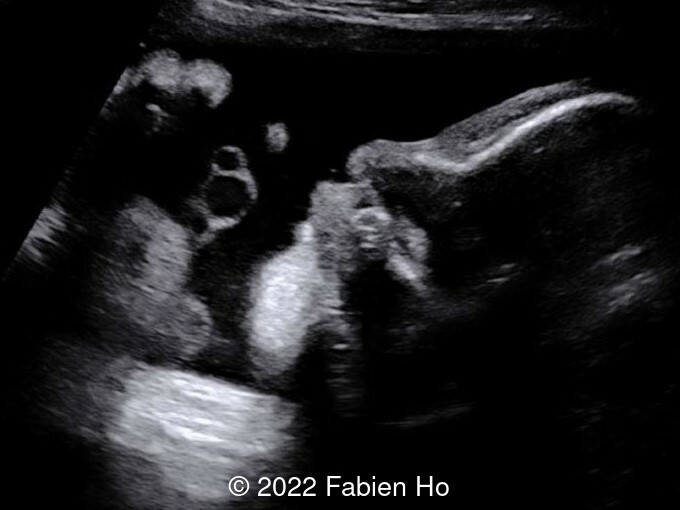
- Image 8: Micropenis with cryptorchidia
- Image 9: Foot malposition
- Image 10 and Image 11: MRI demonstrating brachycephaly, normal brainstem, normal corpus callosum, normal cerebellum and cerebellar vermis, normal brain parenchyma, normal eyeballs, normal choanae. The fetal profile had a frontal-superior angle of 145°, at the upper limit of normal. There are normal semi-circular canals of the inner ears. Absent olfactory bulbs and olfactory sulci (only seen at retrospective review of the MRI postnatally, upper left corner of image 11).
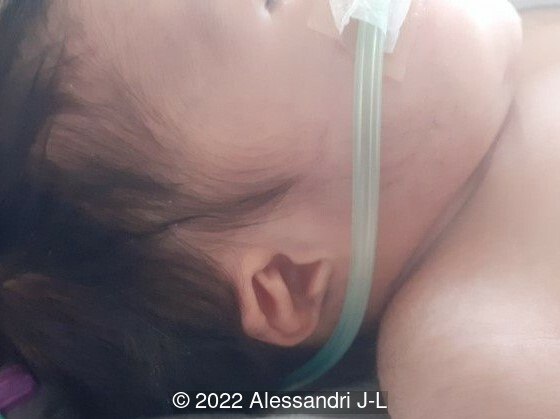
A male neonate was born vaginally at 37 weeks gestation with APGARs 10, 10 and 10. Birth weight was 2400g. Head circumference was 33cm and length was 48cm. Postnatally, the neonate had some respiratory distress due to bronchial congestion.
Clinical examination after birth revealed:
- Sluggish facial expression, malar hypoplasia and nasal bone hypertrophy
- Sagittal craniosynostosis
- Unilateral ptosis
- Dysplasia of the external ear, which were low-set
- Micropenis, small scrotum, left cryptorchidism
- Rocker-bottom right foot
Postnatal investigations included the following:
- Second reading of antenatal MRI demonstrated olfactory bulb agenesis and absence of olfactory sulci
- Neonatal cranial MRI and inner ear CT scan demonstrated olfactory bulb agenesis and abnormal semi-circular canals
- Fundoscopy revealed bilateral coloboma
- Electroencephalogram was abnormal
- Prostigmine test (for congenital myasthenic syndrome) was negative
- Postnatal whole exome sequencing identified a de novo CHD7 gene mutation, confirming our final diagnosis of CHARGE syndrome.
The baby died at 3 months of age due to failure to thrive, hypertension with proteinuric renal failure, and respiratory failure.

Discussion
CHARGE syndrome is an acronym for [1]:
-
Coloboma
-
Heart defect
-
Choanal Atresia
-
Postnatal growth Restriction
-
Genitourinary abnormality and/or hypogonadism
-
Ear anomalies and/or deafness
CHARGE syndrome was originally described in 1979 [2,3] with an incidence of approximately 1 in 8,500 live births [4]. There is a broad phenotype and variable expression of a number of different features [5].
CHARGE syndrome is caused most commonly by a de novo mutation in the CHD7 gene [6]. These mutations are found in approximately 60-80% of patients with a clinical diagnosis [6,7]. Most of these are small, truncating or missense mutations that cause a milder phenotype, which may not be diagnosed prenatally. These mutations are not identified by microarray, but may be identified by whole exome sequencing. Larger truncating lesions are associated with a more severe phenotype with multiple congenital abnormalities which may be diagnosed prenatally. Many of these cases lead to pregnancy loss or termination of pregnancy.
CHARGE syndrome can be diagnosed prenatally, though rare [8,9]. Often it is suspected prenatally and confirmed with molecular testing postnatally [9,10,11]. Isolated or multiple congenital abnormalities are identified prenatally in approximately 45-60% of cases where CHARGE syndrome is confirmed postnatally [7,10]. Prenatal features that may be detectable on imaging include:
-
Cleft lip and/or palate which occurs in 33-50% of patients [5,7].
-
External ear abnormalities, which are present in over 85% of cases and can be assessed on 3D ultrasound [7].
-
Middle ear abnormalities are present in 95% of cases [7]. Hypoplasia of the semi-circular canals may be detected with ultrasound between 20 and 25 weeks of gestation [8,10]. On fetal MRI the semi-circular canal can be evaluated on an axial T2 image in which the main anatomical landmarks include the lower part of the cerebellum in the axis of the cochlea. Due to the limited spatial resolution of fetal MRI, identifying a triangular shaped petrous bone is more straightforward, and this finding is associated with semicircular canal agenesis [12].
-
Arhinencephaly is present in approximately 80% of cases and describes the congenital absence of the olfactory bulbs and olfactory tracts [7]. It can be detected on ultrasound after 30 weeks gestation [8]. On fetal brain MRI, a coronal T2 image allows for the best visualization of these structures [12].
-
Posterior fossa abnormalities occur in approximately 70% of cases and include vermian hypoplasia, partial agenesis of the corpus callosum and ventriculomegaly [7]
-
Congenital heart defects, present in 65-80%, most commonly include atrial septal defects, ventricular septal defects and Tetralogy of Fallot [5,7].
-
Thymic hypoplasia occurs in over 40% of cases [7].
-
Genital abnormalities occur in approximately 90% of males and 15% of females, most often genital hypoplasia including micropenis, hypoplastic testicles, and hypoplastic ovaries [7].
-
Tracheo-oesophageal abnormalities occur in 20% of patients [5,7].
Additional features which are usually only detectable postnatally include:
-
Ocular coloboma, which are present in approximately 70-80% of cases [5,7]. Large ocular coloboma can sometimes be seen on prenatal ultrasound [13,14] and MRI [12]. Often, ocular asymmetry leads to the suspicion of an ocular anomaly. Coloboma can be suspected on fetal MRI when asymmetry of the orbits and distortion of the papilla display a posterior focal bulging of the eyeball on an axial T2 image [12].
-
Choanal atresia is present in approximately 40-50% of cases [5,7]. This can be suspected on prenatal ultrasound [15], and MRI [12]. In the early second-trimester, ultrasound findings may include a transient fluid collection within the obstructed nasal cavity [15]. In the late second and third trimester, the patency of the nasopharyngeal pathway can be shown by using Doppler Color ultrasound in either a coronal or longitudinal sagittal plane of the face during fetal breathing movements [16].
-
Postnatal growth restriction
-
Deafness
-
Developmental delay
-
Growth hormone deficiency
-
Cranial nerve dysfunction
-
Patent ductus arteriosus
According to 2016 diagnostic criteria established by Hale et al, diagnosis of CHARGE syndrome requires the presence of at least 2 major criteria, and any number of other criteria. Major criteria include coloboma, choanal atresia and/or facial cleft, abnormal external, middle, or inner ears, (including hypoplastic semicircular canals), and confirmation of a pathogenic CHD7 mutation on genetic testing [17]. As major criteria are not usually detectable antenatally, prenatal diagnosis is rare.
Legendre et al describe 40 fetuses with CHARGE syndrome and argue that clinical features leading to the diagnosis in fetuses and living affected patients are not the same [18]. Developmental delay, growth retardation and patent ductus arteriosus are only detected postnatally. “Fetal CHARGE” is more likely to present with anomalies of the external ear, hypoplasia of the semi-circular canals, and arhinencephaly [8]. Arhinencephaly and semi-circular canal hypoplasia are highly suggestive of CHARGE syndrome and were present together in 80% of fetal cases [18]. In cases where CHARGE syndrome is suspected, subsequent fetal brain MRI can be pursued starting from the 28th week of gestation to further delineate the anomalies and search for arhinencephaly. The molecular analysis of CHD7 can be used to confirm the diagnosis [7,8,9,10]. It may be postulated that a larger truncating mutation may account for the more severe phenotype which allows prenatal diagnosis.
Differential diagnosis includes 22q11.2 deletion syndrome, Kabuki Syndrome, VACTERL association, Smith-Lemli-Opitz Syndrome and Joubert Syndrome [11].
Neonates with CHARGE syndrome may have several life-limiting medical comorbidities, including congenital heart defects and brainstem dysfunction. In patients who survive beyond the first year of life, blindness and deafness is a major cause of disability [10]. Early diagnosis and coordination of multidisciplinary care is essential.
References
[1] Pagon RA, Graham JM, J Zonana J, et al. Coloboma, congenital heart disease, and choanal atresia with multiple anomalies: CHARGE association. J Pediatr. 1981 Aug;99(2):223-7
[2] Hall BD. Choanal atresia and associated multiple anomalies. J Pediatr. 1979 Sep;95(3):395-8.
[3] Hittner HM, Hirsch NJ, Kreh GM, et al. Colobomatous microphthalmia, heart disease, hearing loss, and mental retardation--a syndrome. J Pediatr Ophthalmol Strabismus. 1979 Mar-Apr;16(2):122-8.
[4] Issekutz KA, Graham JM, Prasad C, et al. An epidemiological analysis of CHARGE syndrome: preliminary results from a Canadian study. Am J Med Genet A. 2005 Mar 15;133A(3):309-17.
[5] Sanlaville, D., & Verloes, A. (2007). CHARGE syndrome: An update. European Journal of Human Genetics, 15, 389–399
[6] Vissers LELM, van Ravenswaaij CMA, Admiraal R, et al. Mutations in a new member of the chromodomain gene family cause CHARGE syndrome. Nat Genet. 2004 Sep;36(9):955-7.
[7] Legendre M, Abadie V, Attié-Bitach T et al. Phenotype and Genotype Analysis of a French Cohort of 119 Patients with CHARGE Syndrome. Am J Med Genet C Semin Med Genet. 2017 Dec;175(4):417-430
[8] Colin E, Bonneau D, Boussion F et al. Prenatal diagnosis of CHARGE syndrome by identification of a novel CHD7 mutation in a previously unaffected family. Prenat Diagn. 2012 Jul;32(7):692-4.
[9] Biard JM, Payrat S, Clapuyt P, et al. Antenatal diagnosis of CHARGE syndrome: Prenatal ultrasound findings and crucial role of fetal dysmorphic signs. About a series of 10 cases and review of literature. Eur J Med Genet. 2021 Apr;64(4):104189.
[10] Busa T, Legendre M, Bauge M et al. Prenatal Findings in Children with Early Postnatal Diagnosis of CHARGE Syndrome. Prenat Diagn. 2016;36(6):561-7
[11] Traisrisilp K, Chankhunaphas W, Sittiwangkul R, et al. Prenatal Sonographic Features of CHARGE Syndrome. Diagnostics (Basel). 2021 Feb 28;11(3):415
[12] Millischer A, Sonigo P, Attie T et al. Fetal MRI Findings in a Retrospective Cohort of 26 Cases of Prenatally Diagnosed CHARGE Syndrome Individuals. Prenat Diagn. 2019;39(9):781-91
[13] Bault JP, Quarello E. Retinal coloboma: prenatal diagnosis using a new technique, the ‘virtual fetal eyeground’. Ultrasound Obstet Gynecol. 2009 Apr;33(4):495-6.
[14] Egloff C, Tassin M, Bault JP, et al. Prenatal description of retinal coloboma. J Gynecol Obstet Hum Reprod. 2020 Sep;49(7):101746.
[15] Bronshtein M, Leibovitz Z, Laham G, et al. Early Transient Prenatal Ultrasound Features of Choanal Atresia. J Ultrasound Med. 2018 Oct;37(10):2439-2444.
[16] Bardin R, Efrat Z, Idelson A et al. Prenatal detection of unilateral nasal airway obstruction caused by a dacryocystocele. Ultrasound Obstet Gynecol. 2016 Feb;47(2):242-3.
[17] Hale CL, Niederriter AN, Green GE, Martin DM. Atypical phenotypes associated with pathogenic CHD7 variants and a proposal for broadening CHARGE syndrome clinical diagnostic criteria. Am J Med Genet A. 2016 Feb;170A(2):344-354
[18] Legendre M, Gonzales M, Goudefroye G et al. Antenatal Spectrum of CHARGE Syndrome in 40 Fetuses with CHD7 Mutations. J Med Genet. 2012;49(11):698-707
Discussion Board
Winners

Javier Cortejoso Spain Physician

Umber Agarwal United Kingdom Maternal Fetal Medicine

Ana Ferrero Spain Physician

Boujemaa Oueslati Tunisia Physician

Murat Cagan Turkey Physician

Ionut Valcea Romania Physician

Dr Monika Sharma India Physician